
|
|
|
CLINICAL STUDIES / ETUDES CLINIQUES
SEIZURE VARIABLES AND COGNITIVE PERFORMANCE IN PATIENTS WITH EPILEPSY
VARIABLE DE CRISES ET PERFORMANCE COGNITIVE CHEZ LES PATIENTS ÉPILEPTIQUES
E-Mail Contact - SUNMONU Taofik A. :
taosunmonu@yahoo.com
ABSTRACT Background and Purpose Methods Results Key words: cognitive, epilepsy, memory, mental speed, Nigeria. INTRODUCTION Epilepsy is the second most common disorder of the central nervous system, affecting 1% of the human population (3). It is particularly highly prevalent in developing African countries (24,28). The concern over the quality of life of patients with epilepsy (PWE) has attracted many attempts at elucidating the impact of seizure variables like seizure types, frequency of seizures, duration of epilepsy, and effect of anti-epileptic drugs (AEDs) on the cognitive functioning of these patients. Several reports have confirmed the presence of cognitive disturbances in PWE and the consequences on their day-to-day activities (1, 9, 20, 29, 30). Cognitive deterioration observed in epilepsy may arise ab initio as a result of the epilepsy as the same brain dysfunction that causes the seizures can also lead to learning disabilities, memory impairment and psychomotor retardation (1,24,28) or it may be secondary to anti-epileptic medications which can exacerbate these cognitive disturbances (9,29). Several reports have shown that cognitive deterioration is worse among patients with generalized seizures than among those with partial seizures (12,15). This explains the contribution of cognitive impairment to poor quality of life among these patients with generalized epilepsies. The longer the duration of epilepsy the higher the likelihood of cognitive deterioration (6, 14) though it has been reported that duration of epilepsy has no effect on cognition (13). Though cognitive disturbances have been noted in newly diagnosed PWE (20, 26), but more importantly is the contribution of the anti-epileptic drugs (AEDs). Anti-epileptic drugs can also contribute to cognitive impairments especially those drugs with significant sedative effects. Phenobarbitone, diazepam, phenytoin, sodium valproate and carbamazepine have all been shown to affect the cognitive abilities of PWE but in differing degrees (9, 29, 31). The effect on cognition is worse with phenobarbitone and diazepam (25, 29). Anti-epileptic drugs (AEDs) may cause impairments in cognitive functions such as attention, memory and psychomotor speed. There is evidence that drug-induced cognitive impairment has great impact on critical daily life function of patients with epilepsy (1,9). The cognitive effects of AEDs are of special concern because they are iatrogenically induced (1). Comparative studies of AEDs in developed countries have shown that while carbamazepine had minimal side effects, phenytoin and phenobarbitone have less favorable cognitive side effects with impairments of mental speed, memory and attention (29, 30,31). These latter two drugs are however the most widely used in developing countries, because of good efficacy, broad spectrum of activity and low cost (10,19). This study was designed to assess the effects of seizure variables, namely; duration of epilepsy, frequency of seizures, seizure types, type of anti-epileptic medication, duration of AED use and age at onset of seizures on the cognitive performances of our patients with epilepsy. A computerized cognitive assessment test battery, the FePsy(16) which has been validated earlier among Nigerians (21) was used to assess the memory, attention, concentration and psychomotor speed of the patients. PATIENTS AND METHODS The cohort comprised of forty-one patients with epilepsy (PWE) and forty-one sex-, age-, level of education- and socio-economic-matched controls. The study participants were adult patients attending the Neurology clinic of Obafemi Awolowo University Teaching Hospitals Complex, Ile-Ife, a tertiary health facility situated in metropolitan Nigeria. Informed consent was obtained from each subject before commencement of the study. The ethical committee of the hospital gave approval for the study. The inclusion criteria included patients aged between 16 and 55years with history of epilepsy as defined by the characteristic eye witness of at least two separate unprovoked non-febrile seizures. Patient must have also completed at least primary school education (some of the test items require subjects to be literate) and must be on phenytoin or carbamazepine therapy only to avoid the confounding effects of polytherapy. These two drugs are the most commonly used anti-epileptic drugs in our center. The exclusion criteria included use of antiepileptic drugs other than phenytoin or carbamazepine, seizures secondary to other causes (such as fever, infection, head injury, cerebrovascular disease), patients with visual and/or hearing impairment, musculoskeletal abnormalities and seizures occurring 24 hours preceding cognitive functions assessment. The controls were age, sex, education and social status matched with PWE and they had no family history of seizures. A questionnaire was developed and applied to all subjects. History was taken from patients and eye witnesses. Emphasis was placed on demographic data (age, sex, level of education) and details of seizure variables which include duration of seizures, seizure types, type of AED administered and duration of drug therapy, age at onset of epilepsy and frequency of seizures. The history of birth, perinatal injury, fever, head injury and drug abuse was sought. Detailed general physical examination was carried out on each subject and the seizure types were classified according to the International League against Epilepsy (ILAE 1981) criteria (21). The cognitive performances of the PWE and controls were assessed with the computerized cognitive assessment test battery, the FePsy (19). Electroencephalographic (EEG) studies were done in all the patients. The differences in sex, age and level of education of the PWE and controls were tested for statistical significance with the chi-square test. The difference in the cognitive performances of the PWE and controls was analyzed using Student t test. The impact of seizure variables on cognitive performances in PWE was analyzed using 2-way Analysis of Variance (ANOVA). The level of significance was taken as p <0.05. RESULTS The study participants comprised 41 PWE and 41 controls with mean ages of 28.32(± 9.22) years and 25.95(± 7.72) years respectively. There were 20 males and 21 females in the PWE group and 26 males and 15 females in the control group. Other demographic details of the study participants are as outlined in table 1. Majority of the PWE had generalized epilepsies (73.2%) with approximately half of this group having secondarily generalized seizures. Complex partial seizures were found to be commoner than simple partial seizures. Most of the patients (65.9%) presented in the second and third decades of life. More than half (51.2%) of the patients who participated in the study had had seizures for more than five years. Sixteen of the patients (39%) had frequent seizures i.e. seizures in excess of more than or equal to one fit in 3-6 months. Carbamazepine was used in most of the patients to achieve seizure control (66.7%). The cognitive performances of the patients with epilepsy (PWE) and the controls are presented in table 3. The PWE had prolonged response time in both auditory and visual reaction tasks when compared to the controls but this difference did not reach statistical significance (p>0.05). There was however a significant difference in the psychomotor speed of the PWE and that of the controls using the complex reaction task (binary choice reaction task). In addition, the concentration ability of the PWE was significantly impaired as shown by their lower percentage of correct responses (p<0.01). The PWE were significantly impaired in their attention span (p=0.005) but not in their recall abilities (p= 0.07). The effects of the seizure variables on the various cognitive tasks performance are presented on table 4. The frequency of seizures and the type of epilepsy did not affect the cognitive performances of the PWE significantly with the exception of their concentration abilities which was negatively impacted by the seizure frequency using the binary choice reaction task (p=0.035). The duration of epilepsy significantly affected the concentration abilities and the verbal memory performance of the PWE negatively (p<0.05) but had no statistically significant effect on other cognitive tasks. The age at onset of epilepsy did not show any appreciable significant effect on their cognitive functioning. The type of medication i.e. AED administered to the patient had no effect on the cognitive performance with the exception of the auditory reaction time which was significantly prolonged using the dominant hand (p=0.014) among PWE on phenytoin. The duration of therapy however significantly affected their reaction time (both auditory and visual), concentration and attention, and their information recall abilities (p<0.05). DISCUSSION Cognitive deterioration in epilepsy has been associated with poor quality of life, marital disharmony and family breakup, inability to secure employment, retrenchment from work, academic disruption and social stigma (5). Most patients with epilepsy report memory impairment during clinic visits as this obviously disrupt their academic performance (20,27). Cognitive dysfunctions which have been associated with epilepsy include memory impairment, psychomotor retardation, inattention and lack of concentration, reduced motor speed and in patients with significant brain damage impaired visual scanning task (1,9,12,20,26). In this study the lack of significant difference in psychomotor and recognition memory performances of the PWE and controls using the auditory and visual simple reaction time tasks is inconsistent with previously documented observations in the literature. This may be as a result of the type of cognitive tests used or the small sample size, though we opined that it is likely due to the latter. A previous publication by Ogunrin et al (20) that studied a larger sample size and used similar cognitive tests showed significant difference between PWE and controls. Despite the fact that the etiology of the epilepsy may cause cognitive disruption by itself, the contributory roles of various seizure variables need evaluation. This study showed inconsistent pattern when effects of seizure frequency and epilepsy type on cognitive performance were assessed. The seizure frequency affected concentration ability but had no effect on mental speed, an observation that is not consistent with most reports in the literature. The lack of effect was noted when the simple reaction tasks were used to assess the (psychomotor) mental speed of the patients. Furthermore, the pattern of performance showed a faster response with the non-dominant hands for the auditory (simple) reaction task, an observation which has been reported earlier and is different with what was observed by researchers in the western countries (20). The negative impact on mental speed was however noted with complex (binary) reaction task. This suggests that more complex cognitive tasks may be more demanding and difficult to cope with in epilepsy. The same pattern was observed for duration of epilepsy and type of medication used. It has been established that the higher the frequency of seizures the more severe the cognitive dysfunctions thus underscoring the importance of effective and optimal seizure control with appropriate AEDs (2,27). Evidences from the literature clearly show that the more frequent the attacks of seizures, the more the likelihood of impaired cognition for both idiopathic and symptomatic epilepsies (1,30). It is likely that recurrent seizures with loss of consciousness directly results in cognitive disturbances by interfering with learning, and indirectly by causing cerebral damage, especially in patients with chronic epilepsy (1,7,12,14). A direct relationship between the number of seizure attacks and cell loss in the hippocampus, an essential structure in some memory processes, has been reported (17). Twin studies have also shown that the twin with more frequent seizures performed worse on tests of cognitive functions (7). The degree of seizure control thus appears to be important as good control has been associated with better performance (18). The age at onset of seizures and the seizure type appeared to have no significant effects on the cognitive abilities of the PWE. This is at variance with earlier observations (6, 14, 15,18). Early onset of seizures is recognized to have deleterious effect on cognition (27). Since childhood is an important period during which one starts learning, then onset of epilepsy during childhood interferes greatly with the acquisition of learning abilities. Many authors have noted that the onset of seizures in early childhood is associated with a higher risk of cognitive impairment than when seizures started in late childhood or the teenage years. Attempts to associate seizure types with cognitive function have not yielded entirely consistent results. Generalised absence seizures have been reported to be less damaging than generalised tonic- clonic seizures, and patients with partial seizures performed better on tests of cognitive tasks than those with generalised seizures (6,27). The cognitive performance was however worse in PWE on phenytoin when compared with those on carbamazepine. This pattern has been consistently noted by other authors (8,23,29,31). The duration of therapy affected all the cognitive tasks assessed portraying the contributory role of prolonged administration of AED on cognitive functioning of PWE. It is important to emphasize that the inconsistencies observed with this study may not be unrelated to the small sample size. Most of the earlier studies among Nigerian Africans assessed larger sample sizes of PWE. Apart from the limitation of small sample size, the other limitation of this study is our inability to assess the serum levels of the AEDs (carbamazepine and phenytoin) in these patients.
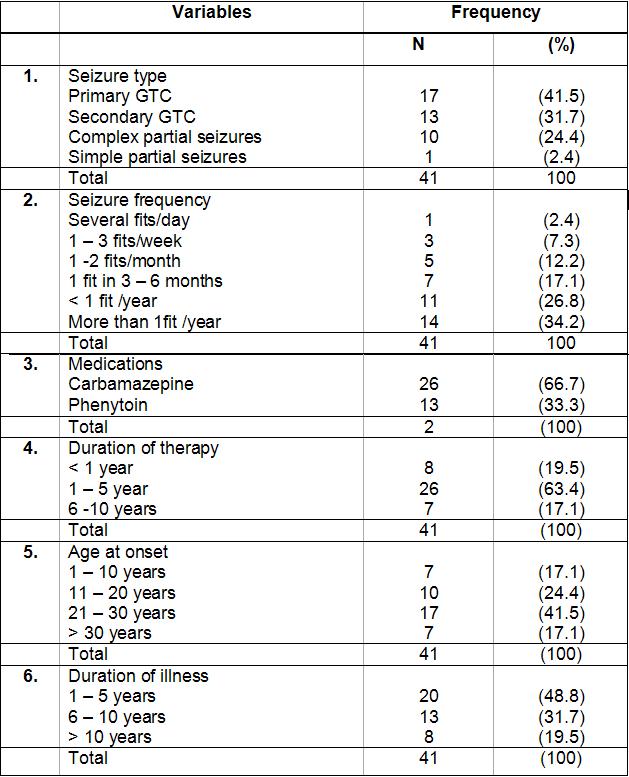 Table 2: Seizure variables in PWE
REFERENCES
|
© 2002-2018 African Journal of Neurological Sciences.
All rights reserved. Terms of use.
Tous droits réservés. Termes d'Utilisation.
ISSN: 1992-2647

