
|
|
|
CASE REPORT / CAS CLINIQUE
PRIMARY CEREBELLAR GLIOBLASTOMA MULTIFORME WITH UNCHARACTERISTIC CLINICAL AND IMAGING FEATURES
GLIOBLASTOME MULTIFORME CEREBELLEUX AVEC DES SIGNES CLINIQUES ATYPIQUES
E-Mail Contact - KITUNGUU Peter Kithikii :
pkitunguu@hotmail.com
ABSTRACT Introduction Case presentation Conclusions Keywords: Cerebellum, Glioblastoma, Posterior fossa RESUME Introduction Présentation de cas Conclusions Mots clés: Glioblastome, Cervelet INTRODUCTION Glioblastomas constitute approximately 15%-20% of all intracranial tumors (6) and generally occur in the fifth and sixth decades. These infiltrating tumors are located in the deep white matter or in the deep gray matter neighboring white matter, mainly in cerebral hemispheres and usually develop secondary to diffuse or anaplasticastrocytomas but can sometimes occur primarily (14)Occurrence of primary cerebellarglioblastoma multiforme (GBM) in adults is extremely rare; few cases have been published so far (3,6,2, 10,13, 9). In this paper, clinical features, imaging and pathologicalfindings of primary cerebellar GBM in a patient are reported. Case Report A 30 yr old female patient, MW, presented with a 3 week history of progressively increasing headaches, vomiting and visual blurring. This was associated with a sudden exacerbation in the headache, loss of consciousness and subsequent progressive coordination deficits. Physical examination was remarkable for diplopia, papilledema and truncal ataxia. Non contrast-enhanced computed tomography (CT) demonstrated a well circumscribed, hemorrhagic lesion in the posterior fossa with little perilesionaloedema. The massenhanced minimally with contrast and compressed the 4th ventricle with resultant dilatation of the proximal ventricular system. Magnetic resonance imaging (MRI) demonstrated a midline cerebellar mass that washyperintense on T1-weighted, predominantly hypointense on T2-wieghted with minimal adjacent edema and hypointense on FLAIR (Figures1-3).It showed blooming on gradient sequence and no restriction with diffusion.The mass had minimal enhancement after administration of gadolinium. 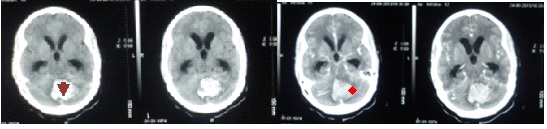 Figure 1: Pre- and post-contrast computed tomography (CT) showing a well circumscribed, hemorrhagic lesion in the posterior fossa (arrow head) with minimal perilesionaloedema and mild contrast enhancement (star). There is associated third and lateral ventriculomegaly 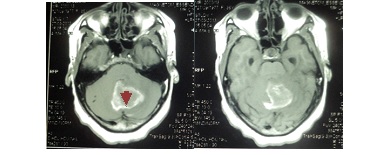 Figure 2: Pre- and post- contrast enhanced T1 weighted MRI images showing a hyperintense midline cerebellar mass with a hypointense core (arrow-head), minimal perilesional edema and mild enhancement with contrast. 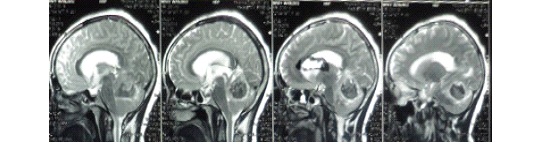 Figure 3: T-2 weighted sagittalimages showing a heterogenous but predominantly hypointenseinfratentorial mass with effacement of the 4th ventricle and proximal ventriculomegaly The patient underwent suboccipital craniotomy and gross total resection of the mass was achieved. Pathologic examination revealed a highly cellular tumor with marked nuclear atypia, numerous mitoses and areas of necrosis. There was considerable nuclear and cytoplasmicpleomorphism, with multinucleated, giant cells and hemosiderin-laden macrophages (figures 4-5). 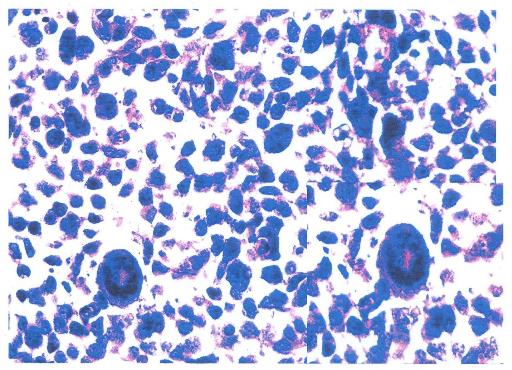 Figure4: Photomicrograph showing a highly cellular tumor with marked nuclear atypia, numerous mitoses and multinucleated giant cells (inset). 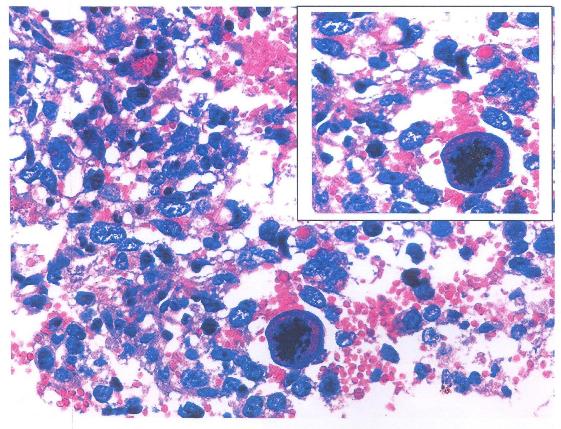 Figure 5: Photomicrograph demonstrating nuclear and cytoplasmicpleomorphism, features of intratumoral hemorrhage and multinucleated giant cells (inset). The post-operative course was uneventful with the patient being admitted in the neuro-intensive care unit for overnight observation and discharged on the 7th post-operative day through the radiotherapy unit for whole-brain radiotherapy.The patient is on follow-up at our out-patient neurosurgical clinic for 7 months with a good outcome and no recurrence, however close monitoring is being observed. DISCUSSION Glioblastoma, the most frequent tumor among all primarytumors of the central nervous system in adults,has a frequency of 50% (2,13). However, adult cerebellarGBM is extremely rare, accounting for 0.24% to 3.8% ofall intracranial glioblastomas( 2,3,7,9,10,13). From 1975 to2011, 170 articles and abstracts about cerebellum glioblastomawere published, according to a search of theMedline database. The male-to-female ratio is 2:1 (7). Cerebellarglioblastoma can be seen in all age groups. About 70% of lesions occur in adults with a medianage of 46.7 years while 30% were noted in children (Demiret al2005; Mattoset al2006). As with our patient,localization is generally median or paramedian with apossible extension to the fourth ventricle (7). The tumoris infiltrative and usually is localized in the deep whitematter (7). The clinical features of patients with cerebellar GBM are similar to those of other aggressive fast growing infratentorial tumors. Signs and symptoms include headache, nausea, vomiting, and cerebellar dysfunction including ataxia, imbalance and unsteady gait (1,4,10,12). Non-enhanced CT scan findings of GBM may include a heterogeneous poorly marginated mass; internal areas of low or fluid attenuation that are the foci of necrosis (present in as many as 95% of GBMs); internal areas of high attenuation that are the foci of hemorrhage or, rarely, calcifications. There may be significant mass effect and perilesionaledema.Enhanced CT scans display significant enhancement with findings such as irregularity and heterogeneity. In contrast, for our patient, the non contrast-enhanced computed tomography (CT) demonstrated a well circumscribed, hemorrhagic lesion in the posterior fossa with minimalperilesionaloedema and did not enhance avidly post contrast administration. The imaging features of cerebellar GBM are described as nonspecific ( 2,7,13). Lesions may occur laterally in the cerebellar hemispheres or in the midline within the vermis. The lesions are typically infiltrating with indistinct margins. Signal characteristics are heterogenous, often with necrotic and cystic components. A thick and irregular wall is commonly seen. However, irregular peripheral enhancement is consistently described following contrast administration. Edema is usually present and obstructive hydrocephalus is common (2,7,13). This is in contradistinction to the imaging findings in our case where the features of perilesional edema and contrast uptake were sublte. Additionally, MRI has a highest degree of confidence in the diagnosis of glioblastomamultiforme (GBM; malignant glioma). MRI findings demonstrate a heterogeneous mass that is generally of low signal intensity on T1-weighted images and high signal intensity on T2-weighted images.There are internal cystic areas, areas of high signal intensity on T1 (hemorrhagic foci), neovascularity, necrotic foci, significant peritumoralvasogenic edema, and significant mass effect. Irregular but intense enhancement after the administration of gadolinium-based contrast material (same pattern as with enhanced CT scanning) is also found (2,7). However, the patient being presented did not conform to these described findings. Magnetic resonance imaging (MRI) demonstrated a midline cerebellar mass that was hyperintense on T1 with minimal adjacent edema, blooming on gradient sequence and no restriction with diffusion. The mass had minimal enhancement with gadolinium contrast. The histology and biology of cerebellar GBM is similar to that of cerebral GBM [3]. This includes malignant tumor cells, mitoses, hypercellularity, pleomorphism and neoangiogenesis. The presence of necrosis helps differentiate GBM from anaplasticastrocytoma or from well-differentiated astrocytoma (Luccarelliet al 1980; Georges et al 1983; Katz et al 1995; Rizket al 1994). The case being presented exhibited these features as well as considerable nuclear and cytoplasmicpleomorphism, with multinucleated, giant cells and hemosiderin-laden macrophages. As with any GBM and any malignant brain tumor, cerebellar GBM has a very poor prognosis. This is attributed to rapid tumor progression, locally aggressive behavior as well as the common findings of CSF pathway spread (8). Early intervention including aggressive surgery as well as aggressive radiation and chemotherapy (2,5,13,16) have been advocated to increase the disease free interval and to prolong survival. Despite these measures; however, survival of patients with cerebellar GBM is very poor, in the range of 3-22 months (2,13). This case is an unusual presentation due to the presence of significant hemorrhage, well-defined margins, minimal contrast enhancement and minimal edema. There were few features helpful in making the correct specific prospective diagnosis of glioblastomamultiforme. However, GBM should be included in the differential diagnosis of a hemorrhagic infratentorial mass with rapid progression of clinical findings as well as imaging findings considered atypical for the common entities that occur in the posterior fossa. REFERENCES
|
© 2002-2018 African Journal of Neurological Sciences.
All rights reserved. Terms of use.
Tous droits réservés. Termes d'Utilisation.
ISSN: 1992-2647