CASE REPORT / CAS CLINIQUE
GÉLINEAU SYNDROME: LITERATURE REVIEW AND MANAGEMENT OF A NIGERIAN TEENAGER
SYNDROME DE GELINEAU. A PROPOS D'UN CAS NIGERIAN ET REVUE DE LA LITERATURE
- Neurology Unit, Dept. of Medicine, UCH, PMB 5116, Ibadan, Oyo State, Nigeria
SUMMARY
Gélineau syndrome is a rare disorder with a hospital prevalence of 0.026% in Nigeria. A 14-year old Nigerian with narcolepsy, cataplexy, visual hypnagogic hallucinations and hypnapompic sleep paralysis is presented here. He had a Multiple Sleep Latency Test done using the Carscadon protocol. With the exclusion of other differential diagnoses, this patient satisfied the DSM IV, Carscadon’s Multiple Sleep Latency Test and the Silber Category A criteria for definite narcolepsy. He was controlled on methylphenidate and programmed daily naps.
Keywords : Cataplexy, Narcolepsy, Nigeria
RESUME
Au Nigéria, le syndrome de Gélineau est un trouble rare avec une prévalence hospitalière de 0.026%. Nous étudions ici le cas d’un patient nigérien âgé de 14 ans atteint de narcolepsie, de cataplexie, d’hallucinations hypnagogiques visuelles et de paralysie du sommeil hypnopompique. Il a été effectué un test de latence multiples d’endormissement en utilisant le protocole de Carscadon. En excluant d’autres diagnostics différentiels, ce patient répond au DSM IV, le test de latence multiple d’endormissement de Carscadon et du Silber Categori un critère déterminant la narcolepsie. Le patient a été mis sous méthylphénidate et des siestes quotidiennes lui furent préconisées.
Mots clés: Afrique, Cataplexie, narcolepsie, Nigéria
INTRODUCTION AND LITERATURE REVIEW
The term narcolepsy is derived from Greek narke stupor, lambanein : to seize, and literary means seized by somnolence’. Gelineau was the first to describe the syndrome in 1880. Narcolepsy is characterised by the classic tetrad of excessive daytime sleepiness, cataplexy, hypnagogic hallucinations and sleep paralysis.(4, 10,12,15)
However, the diagnostic criteria, though debatable, has continued to evolve from 1880 through 1975 [the First International Symposium on Narcolepsy] till date.(2,4,19)
It is a rare disorder with an incidence of 0.02 to 0.07% in North America, 0.002% in Israel, 0.04% in United Kingdom. ( 5,12) In Nigeria, the hospital prevalence rate is 0.026 %20 Males are affected somewhat more often than females. ( 5,12) Age at onset varies from the second to the fifth decade with the highest peak at 15 years and a less pronounced peak at 36 years. Onset of idiopathic narcolepsy is before the age of 30 years in about three quarters of the cases.(5) Narcolepsy has been reported in children as young as 2 years. (10) First degree relatives have a 10 – 60 fold higher risk than the general population.(5,10)
The aetiology of narcolepsy is still unknown. It is most frequently a sporadic disorder resulting from genetic and environmental factors. (4) Narcolepsy-cataplexy is strongly associated with HLA-DR2, and 85-98% of Caucasian patients are DR2 positive.(10) In black populations, this is less so. HLA DQB1*0602 is associated more strongly with narcolepsy-cataplexy [76%] than with narcolepsy without cataplexy. HLA DQA1* 0102 is also associated with the condition.
A recent development in the pathogenesis of narcolepsy is the identification of an abnormality in the hypocretin [orexin] receptor 2 gene in the canine model.(10) Low CSF hypocretin is highly specific [99.1%] and sensitive [88.5%] for narcolepsy – cataplexy.5 The hypocretins /orexins are hypothalamic peptides most recognised for their effects on feeding and arousal.(11) Autoimmune mediated degeneration of hypocretinergic systems in the lateral hypothalamus has been hypothesised as a possible aetiology for the syndrome.
Also implicated in the pathogenesis of narcolepsy are up-regulation of type 2 muscarinic receptors in the pontine reticular formation, involvement of alpha 1 and 2 noradrenergic receptors, dysfunction of the meso-cortico-limbic dopaminergic system and possibility of autoimmune and cytokine [TNF -alpha] damage. (6,10,18)
Secondary/symptomatic forms have been rarely described following head trauma [as in a patient that we recently managed, and being presented in this communication], in encephalitis lethargica, multiple sclerosis and meso-diencephalic vascular, neoplastic and degenerative lesions. (4,5,12,15)
The clinical features of narcolepsy syndrome can be viewed as rapid eye movement (REM) sleep components intruding into wakeful states or light sleep. This is because the monoamine-dependent inhibition of cholinergic REM-on cells may be defective. (10,14,15) Cataplexy and sleep paralysis represent intrusion of REM sleep atonia, whereas hallucinations represent an intrusion of dreams. (10)
CASE REPORT
OA is a 15 year old Senior Secondary School student who presented with excessive daytime sleepiness of 4 months duration. He had sleep attacks consisting of sudden and deep sleeps in unusual circumstances such as while walking, eating, ironing, writing examination, talking and performance of daily chores. He had 5 -10 naps per day. He had visual hypnagogic hallucinations, which he described as continuation of visual experience of wakefulness into sleep and fall into dreamy state immediately he falls asleep. He also had hypnapompic sleep paralysis which he expressed as feeling of somebody pinning him down’ anytime he attempts to rise up from the bed. He occasionally experienced speech arrest/paralysis and cataplectic attacks. These were described as feeling of sudden severe generalised body weakness in full consciousness, while walking, standing, laughing and/or during excitement. Even though he feels very weak, he does not actually fall to the ground. Each episode lasts about 1-5 minutes.
He sleeps between 10pm to 7am though he experiences interruptions as he wakes up about three times during the night. As reported by his parents, he neither snores nor sleepwalks and experienced no period of cessation of breathing, nightmare, panicky dream or abnormal limb movements during sleep. He had no fever, headache, seizures, nausea, vomiting, hearing or visual impairment or dizziness. He was not on any drug and neither smokes nor drinks alcohol. He had no symptom suggestive of organ failure, endocrinopathy or metabolic derangement and his appetite for food remained the same. He had scalp laceration at the age of ten when he fell into a ditch without impairment of consciousness, vomiting or headache. He did not have features such as poor concentration, impaired memory or tiredness/fatigue that are more prominent in post-concussion syndrome. (14) Pregnancy, birth, neonatal and childhood history were otherwise uneventful. He had no family history of sleeping disorder. He is the 3rd of 5 children in a monogamous family.
Physical examination revealed a young boy with a body mass index [BMI] of 17.6kg/m2. Comprehensive clinical assessment of his general state, higher mental functions, cranial nerves, motor, sensory and cerebellar functions revealed no abnormality. His occipito-frontal circumference was 56cm. Detailed cardiovascular, respiratory and abdominal examinations were normal.
His packed cell volume was 38%, total white cell count 4,300/mm3, neutrophil 34%, lymphocyte 58%, monocyte 8%; platelets adequate. The erythrocyte sedimentation rate was 8mm/hr by Westergren method. The clinical impression was narcolepsy. The absence of fever, wasting, lymphadenopathy and hepatosplenomegaly made trypanosomiasis unlikely, while the non-periodic EDS excluded Kleine-Levin’s recurrent hypersomnia syndrome (14)
A multiple sleep latency test was performed 6 months after presentation without any stimulants, and while maintaining his usual sleep schedule.(13) The findings are shown in table 1. An episode of REM desynchronisation is shown in figure 1. The Carscadon protocol was followed.(5,8,20) The patient was given 20-minute opportunities to fall asleep on a comfortable bed in a dark, cool and quiet room. Whenever he fell asleep, he was not allowed to sleep for more than 15 minutes in any session. The periods from lights out to sleep onset [sleep latency period] were charted while his electroencephalogram was recorded from lights out to 5-15 minutes after sleep onset. The test was repeated at 2-hourly intervals. He had no epileptiform features on EEG. With 3 sleep-onset REM periods [REM low-voltage fast frequency alpha desynchronisation occurring within 15 minutes of sleep onset] and an average sleep latency period of 2.8 minutes, he fulfilled the EEG criteria for narcolepsy. He was asked to take short programmed naps during the day and 10mg of methylphenidate daily. He reported no side-effects and his performance of daily and school activities improved remarkably.
DISCUSSION
Narcolepsy is a distinctive neurological syndrome with two major and two minor symptoms. The two major symptoms are excessive daytime sleepiness and cataplexy, while the minor symptoms are hypnagogic hallucinations and sleep paralysis.
Excessive daytime sleepiness [EDS] is the primary symptom of narcolepsy. It is usually the first symptom and is present in all patients with narcolepsy. Severe EDS as experienced by this patient leads to involuntary somnolence during more active conditions like eating and talking.7 For instance, the index patient had sleep attacks occurring without warning while ironing and writing examinations. Each attack lasts about 10 -30 minutes; and the individual has 2-6 attacks per day, though some may have up to 30 attacks per day.(15) Attacks are usually refreshing, with a refractory period of one to several hours before the next episode occurs.(10,12,15) In females the severity of attacks could be affected by the menstrual cycle.(15) Apart from irresistible sleep episodes, patients may have memory lapses, drowsiness, speech and gestural deambulatory automatisms.(12)
It is noteworthy that the total amount of time spent in REM sleep is normal. Hence excessive sleep during the day leads to shorter and disturbed deep sleep at night.(15) In fact, continuous polygraphic monitoring of narcoleptics in time isolation [up to 19 days] showed that the total fraction of time spent asleep and sleep stage composition were no different from normal controls.(5,17)
Cataplexy – astasias [Latin, to strike down with fear’] is an abrupt attack of muscle weakness seen in about 70% of narcoleptics and preceding EDS in 2 -3 % of cases. (5,10,12,15) It could be generalised or segmental.(5) It is an abrupt and reversible decrease or loss of muscle tone, most frequently elicited by strong emotion: laughter and anger; and less often: excitement, surprise, heavy meals, sudden exertion, reading, listening to music or remembering an emotional event. (5,10,12,15) There may not be any clear precipitating act or emotion. It is associated with REM-sleep related suppression of monosynaptic H-reflex and multi-synaptic tendon reflex activity leading to atonia and areflexia.(12) This is in spite of preservation of clear consciousness in most patients, though rarely, concurrent EDS may lead to sleep in a patient with severe total body cataplexy.(5)
The frequency, severity, extent and duration of cataplexy are widely variable. (5,10,12,15) It could present with minimal weakness or absolute powerlessness; segmental involvement of facial muscles/palpebral muscles/ muscles of speech/jaw muscles/neck muscles/abdominal muscles/ knee extensors or total body weakness; fleeting or prolonged attack lasting 15-45 minutes[ status cataplecticus.( 5,10,12) The patient being discussed had occasional episodes of speech arrest and transient total body weakness without falls following laughter or excitement.
Hypnagogic and/or hypnapompic hallucinations and/or sleep paralysis occur in one sixth to one-third of patients. The hypnagogic hallucinations are often visual, usually consisting of simple forms that are constant or changing in size. Vivid auditory or cenesthopathic hallucinations may also occur with out of body experiences. In sleep paralysis the patient finds himself unable to perform any voluntary action despite full awareness. Patients experience frightening difficulties to move the limbs, open the eyes or even speak. (5,10,12,15) Episodes rarely lasts more than 10 minutes and resolve spontaneously or with tactile stimulation. Sleep paralysis occurs in healthy subjects during REM sleep. (5,10,12,15) Hypnogogic and/or hypnopompic hallucinations , sleep paralysis and cataplexy tend to improve with age( 5,10,12,15).
The index patient had both hypnogogic hallucinations and hypnopompic sleep paralysis.
No specific physical finding on examination suggests narcolepsy, though some patients may be obese (10).
The diagnostic criterion of narcolepsy is in constant evolution.(4) The First International Symposium on Narcolepsy 1975 defined it as a syndrome of unknown origin that is characterized by abnormal sleep tendencies, including excessive daytime sleepiness and often disturbed nocturnal sleep and pathological manifestations of REM sleep. The REM sleep abnormalities include sleep-onset REM periods and the dissociated REM sleep inhibitory processes, cataplexy, and sleep paralysis. Excessive daytime sleepiness, cataplexy, and less often sleep paralysis and hypnagogic hallucinations are the major symptoms of the disease.
Guidelines for the performance of Multiple Sleep Latency Tests [MSLT] was established by Carskadon et al in 1986 and still remains the best available technique for confirming the diagnosis of narcolepsy .(5,8,20) Briefly, the patient goes to bed for four to six 20-minute opportunities to sleep, spaced 2 hours apart, generally beginning at 10am. After each episode the patient stays awake until the next. The patient is not allowed to sleep for more than 15 minutes per episode. During each episode the latency between lights-out time and sleep-onset and REM sleep is determined. Sleep onset REM period SOREMP is defined as REM sleep that occurs within 15 minutes of sleep. REM sleep is seen on EEG as fast low voltage alpha rhythm (5, 8,12,).
To be valid, the patient should be off stimulants and REM suppressant drugs for at least 2 weeks and should have had his /her usual night’s sleep on a fairly unvarying schedule for at least 1 week before the test. Certain sleep labs also do a polysomnography the night before and a urine drug screen on the day of the test (5,8,12,13).
Two or more SOREMPs and an average sleep latency of less than 5 minutes is required to confirm the diagnosis.(7) However , 3 or more SOREMPs with a mean sleep latency of less than five minutes is 99.2% specific for and 87% positively predictive of narcolepsy (10).
The DSM-IV 1994 diagnostic criterion requires daily irresistible attacks of refreshing sleep over at least 3 months; and cataplexy and/or hypnapompic/hypnagogic hallucinations/sleep paralysis and exclusion of other disorders (9).
M.Silber et al in 2001 developed a classification of narcolepsy into 4 categories. Category A [Definite Narcolepsy] : objectively confirmed sleepiness [EDS], cataplexy, 2 or more SOREMPs, and exclusion of other disorders. Category B [Probable Narcolepsy- Laboratory Confirmation]: EDS objectively confirmed and either cataplexy [category B1] or 2 or more SOREMPs [category B2]. Category C [Probable Narcolepsy – Clinical]: a history of EDS and cataplexy but without laboratory studies (18,19).
This patient satisfied the DSM IV, the MSLT and the Silber category A criteria for definite narcolepsy since other disorders were excluded. Even though SOREMPs may occur in depression, severe obstructive sleep apnea, sleep deprivation and recent withdrawal from REM – suppressant drugs; the presence of cataplexy, hypnagogic hallucinations and sleep paralysis effectively rules out these and other hypersomniac disorders in this patient who belongs to the susceptible age group and sex for narcolepsy. (5,12,15) Idiopathic CNS hypersomnia is excluded by the presence of SOREMPs. (12,14)
Recently Bassetti et al proposed a multimodal approach to diagnosis including low CSF hypocretin-1 [orexin A, 99.1% specific and 88.5% sensitive], HLA-DQB1* 0602 positivity, high CSF leptin and high BMI . (2) This however is not yet generally acceptable and available. Neuroimaging is normal in narcoleptics and is only required to confirm the suspicion of structural lesions (10).
Therapy is both pharmacological and non-pharmacological.
Pharmacological therapy is detailed in table2. Non-pharmacological approach includes 2 or 3 scheduled 15- to 20-minute daytime naps with resultant variable 90- to 120-minute refractory sleep period. Emotional support, counselling; psychosocial, vocational therapies are also useful. Sleep hygiene and after-school exercise programmes are usually beneficial (4,5,10,12,14,15).
Our patient responded well to methylphenidate 10mg daily, and 2-3 scheduled naps with improved performance of daily chores and school activities.
TABLE 1: Results of Multiple Sleep Latency Tests performed for OA on 14/10/03 in UCH EEG Laboratory
| Session |
Sleep latency period [minutes] |
Sleep duration [minutes] |
SOREMP latency period |
| 1 |
5 |
5 |
|
|
| 2 |
2 |
5 |
1 minute 28 seconds |
| 3 |
1 |
9 |
5 minutes 23 seconds |
| 4 |
3 |
10 |
|
|
| 5 |
3 |
5 |
2 minutes 22 seconds |
TABLE 2 : Pharmacological treatment of narcolepsy-cataplexy (3-5,10,12,14,15)
| Drug |
Posology |
Classification |
Side effects |
| Dextroamphetamine, Amphetamine, Methamphetamine |
10mg bid to 40mg/day |
CNS stimulant : for EDS |
Sympathomimetic[tachycardia, hypertension] , Mood – altering [irritability, lability], Cognitive [racing thoughts], tolerance, dependence, headache, GIT side-effects, sleep disturbance |
| Methylphenidate [ Long considered drug of choice by most] 1,3 |
10mg bid to 20mg qid |
CNS stimulant : for EDS |
As above but lower side effects |
| Pemoline |
18.75mg to 150mg in single or divided doses [bd] |
Less potent amphetamine, for EDS |
Sympathomimetic[tachycardia, hypertension] , Mood – altering [irritability, lability], Cognitive [racing thoughts] , tolerance, dependence, headache, GIT side-effects, sleep disturbance,hepatic dysfunction. |
| Mazindol |
< 5mg/day |
-ditto- |
-ditto- |
| Modafinil [novel drug, licensed in 1999] |
200-400mg/day |
Alpha-1 receptor agonist, activates orexin-containing neurons |
Headache |
| Imipramine, Clomipramine Desipramine |
< or= 200mg/day |
Tricyclic antidepressant for REM related phenomena |
Atropinic side-effects |
| Protryptiline |
< or= 20mg/d |
-Ditto- |
-Ditto- |
| Fluoxetine |
|
Selective Serotonin Reuptake Inhibitor for REM related phenomena |
Sleep disturbance, sexual dysfunction |
| Viloxanine |
< or= 200mg/day |
Norepinephrine Reuptake Inhibitor for REM related phenomena |
|
|
| Codeine |
30mg 4hrly-up to 150mg/day |
|
|
|
| Gamma-hydroxybutyrate, |
1g twice nocte |
Improves night sleep |
|
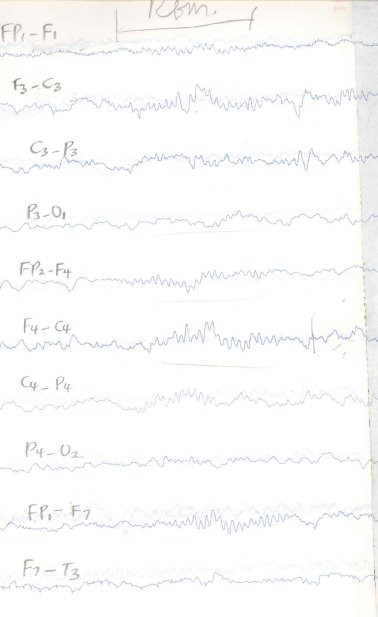 Figure 1
| ACKNOWLEDGEMENT |
| We wish to acknowledge the efforts of Mr. Molade the EEG Technologist who recorded the EEG. |
REFERENCES
- BADEMOSI O, KUKU A, ADEUJA AOG, OGUNNIYI A, OSUNTOKUN BO. Narcolepsy in Africans : A clinical study of 23 Nigerian subjects. Afr. J. Neurol. Sci. Vol1990 ;9: 5-8
- BASSETTI C, GUGGER M., BISCHOFF M, MATHIS J, STURZENEGGER C, WERTH E, RADANOV B, RIPLEY B, NISHINO S, MIGNOT E. The narcoleptic borderland: a multimodal diagnostic approach including cerebrospinal fluid levels of hypocretin-1 [orexin A] Sleep Med . 2003;4[1]:7-12.
- BECKER PM, SCHWARTZ JR, FELDMAN NT, HUGHES RJ. Effect of modafinil on fatigue, mood and health-related quality of life in patients with narcolepsy. Psychopharmacology [Berl]. 2003 Nov 25. [Epub ahead of print]
- CZEISLER CA, WINKELMAN JW, RICHARDSON GS. Sleep disorders. Fauci AS, Braunwald E et al. editors. In Harrison’s Principles of Internal Medicine. 15th edition. Singapore. Mc Graw-Hill Book Co.2001: Section 3 ;27
- DANIEL RW. Sleep and arousal disorders. In Rosenberg RN. Pleasure DE. Editors. Comprehensive Neurology. New York. John Wiley and Sons. 1998;511-544.
- DAUVILLIERS Y. [Neurodegenerative, autoimmune and genetic processes of human and animal narcolepsy] Rev. Neurol [Paris]. 2003 Nov;159 [11 suppll]:83-7
- DAUVILLIERS Y. , BAUMANN C, CARLANDER B, BISCHOF M, BLATTER T, LECENDREUX M, MALY F, BESSET A, TOUCHON J, BILLIARD M, TAFTI M, BASSETTI CL. CSF hypocretin -1 levels in narcolepsy, Kleine-Levin syndrome, and other hypersomnias and neurological conditions. J. Neurol Neurosurg Psychiatry. 2003; 74[12]:1667-1673
- DEMENT WC, MITLER MM, ROGH T, CARSKADON MA Guidelines for the multiple sleep latency test [MSLT]: a standard measure of sleepiness. Sleep 1986; 9: 519-524.
- DSM IV diagnostic criteria for narcolepsy. American Psychiatric Association: Diagnostic and Statistical Manual of Mental Disorders, ed 4. American Psychiatric Association, Washington, 1994.
- emedicine.com accessed on 15th March 2002.
- FERGUSON AV, SAMSON WK.The orexin/hypocretin system: a critical regulator of neuroendocrine and autonomic function. Front Neuroendocinol. 2003;24[3]:141-50
- GUILLEMINAULT C. Narcolepsy syndrome. In Kryger MH, Roth T, Dement WC. Editors. Principles and Practice of Sleep Medicine. Philadelphia. W.B. Saunders Company.1989:338-350
- JANJUA T., SAMP. T, CRAMER-BORNEMANN M, HANNON H, MAHOWALD MW. Clinical caveat: Prior sleep deprivation can affect the MSLT for days. Sleep Med. 2003;4[1]:69-72
- JUNE MF. Sleep disorders. In Rowland LP. Editor. Merritt’s Neurology.Philadelphia. Lippincott Williams and Wilkins. 2000.pp839-846
- MARSDEN CD Narcolepsy and related sleep disorders. In Weatherall DJ, Ledingham JGG, Warrel DA . Editors. Oxford Textbook of Medicine. Oxford. Oxford University Press. 1995;24:4.3
- PARKES JD. Disorders of sleep. In Bradley WG, Daroff RB, Fenichel GM, et al. Editors. Neurology in clinical practice. Boston.Butterworth-Heinemann
1991;1479-1506
- POLLAK CP, MOLINBE ML, WAGNER DR. Sleep times in narcoleptic subjects isolated from time cues. Sleep Res. 16:406.
- SILBER M., KRAHN L., OLSON E. Diagnosing narcolepsy: validity and reliability of new reseach criteria. JNS 2001;187:suppl 1, S432.
- SILBER MH , KRAHN LE, OLSON EJ. Diagnosing narcolepsy: validity and reliability of new diagnostic criteria.Sleep Med. 2002;3[2]:109-13.
- WILLIAMS A., HIGGINS SE, HOWARD RS. Assessment of sleepiness in the clinic population- The value or not of the MSLT. JNS 2001;187: Suppl 1, p S531.
|
|

