
|
|
|
CLINICAL STUDIES / ETUDES CLINIQUES
A RETROSPECTIVE ANALYSIS OF THE EFFICACY AND SAFETY OF BOTULINUM TOXIN IN THE TREATMENT OF THE MAIN FOCAL DYSTONIAS AFFECTING THE HEAD AND NECK
ANALYSE RÉTROSPECTIVE DE L'EFFICACITÉ DE LA TOXINE BOTULIQUE DANS LE TRAITEMENT DES DYSTONIES FOCALES AFFECTANT LA TÊTE ET LE COU
E-Mail Contact - BHOLA Sudika :
sudikabhola@gmail.com
ABSTRACT Background This study sought to analyse the treatment, duration of benefit, most frequently injected muscles, complications and outcomes of patients receiving botulinum A toxin injections. Methods Results Keywords: Toronto Western Spasmodic Torticollis Rating Scale, Focal dystonia, Botulinum A toxin INTRODUCTION Dystonia is a hyperkinetic movement disorder characterised by patterned, directional and sustained muscle contraction that produces abnormal postures or repetitive movements.2,9 Focal dystonia (FD) is the commonest form of dystonia and can involve any body part. 13 Commonest focal dystonias include blepharospasm, hemifacial spasm and cervical dystonia. Botulinum toxin is a potent biological neurotoxin that produces temporary muscle weakness by causing the irreversible presynaptic inhibition of acetylcholine release.8 It has become a powerful therapeutic tool for a growing number of clinical indications and is the mainstay of treatment for focal dystonia and focal limb dystonia.4,8 In addition to involuntary movement disorders, botulinum toxin has been used effectively to treat disorders of muscle tone, including spasticity associated with cerebral palsy, strokes, brain trauma and multiple sclerosis.4 The aim of this study was to describe the local experience with the use of botulinum toxin injections for these indications. SUBJECTS AND METHODS A retrospective longitudinal study of patients receiving botulinum toxin for hemifacial, spasm, cervical dystonia, and blepharospasm was done. The study was approved by the Bioethics Research Committee at the University of KwaZulu-Natal, Durban (Reference No. BE023/14). 70 medical records of first time patients attending the IALCH botulinum toxin clinic between the 1st January 2003 to 31st July 2012, were reviewed. Patients with diagnoses other than cervical dystonia, hemifacial spasm or blepharospasm or those with incomplete data were excluded. Data was recorded on a structured format which included gender, age and race, diagnosis, dose of botulinum toxin injected, response to therapy, duration of benefit (weeks), complications of therapy, muscles injected and the experience of the injectors. The response of patients was recorded as good response (no spasm), partial response (mild spasm) or no response. The specific type of botulinum toxin A used in this study was Botox. The duration of benefit was in weeks and the experience of the injectors was also recorded to assess whether fewer years of experience correlated with a poorer response. In the cervical dystonia group, the dose at first and last visit was correlated with the Toronto Western Spasmodic Torticollis Rating Scale (TWSTRS) to ascertain if a change in dose led to a better response. The TWSTRS is a composite scale which covers different features of cervical dystonia. The first part is based on the physical findings (severity subscale), the second part rates disability, and the third part pain. The TWSTRS-Severity scale includes the following items: A. maximal excursion (rotation, tilt, anterocollis or retrocollis, lateral shift, sagittal shift), B. duration factor, C. effect of sensory tricks, D. shoulder elevation/anterior displacement, E. range of motion (without the aid of sensory tricks), F. time (up to 60s that the patient is able to maintain the head within 10 degrees of the neutral position without the use of sensory tricks). The sum of A to F amounts to a maximum score of 35 with the duration factor weighed twice. The TWSTRS-Disability is a six-item scale that comprises an assessment of performances of daily activities which may be affected by cervical dystonia: work performance (job or domestic), activities of daily living (feeding, dressing, hygiene), driving, reading, watching television and leisure activities outside the home. Each item is rated on a 6-point scale (0=no difficulty, 5= highest degree of disability). The TWSTRS-Pain consists of a severity score for the patient’s usual, worst and best pain in the last week, as well as a duration component and an assessment of the contribution of pain to disability with a score range between 0 and 20. 6 STATISTICAL METHOD Demographical data was expressed using descriptive statistics. Categorical variables were analysed using the Chi-squared test. The unpaired T test was used for comparisons. A p<0.005 was considered as significant. RESULTS Figure 1 shows the breakdown of patients included in the study. During the study period, 70 patients were included. The demographic data of patients are presented in Table 1. Eight (11.4%) patients had blepharospasm, 28 (40%) had hemifacial spasm and 34 (48.6%) had cervical dystonia. The women were older than men (65.9±13.7 versus 55.6±4.4 years; p<0.003). The doses of botulinum toxin required to elicit an optimal response are shown in Table 1. The mean doses of botulinum toxin injection in females were 23.3 ± 12.1 units for blepharospasm, 22.5 ± 5.2 units for hemifacial spasm and 83 ± 28 units for cervical dystonia. The respective male doses were 27.5 ± 10, 22.6 ± 4.3 and 80.6 ± 26 units. There was no significant difference in dosing units of botulinum A toxin between male and female patients. The response of patients to botulinum toxin injections is expressed in Table 1. A good response was seen in the majority of patients. There was a wide variation in the duration of relief from symptoms among patients with either disorder. The mean duration of benefit with botulinum A injection was 4.5±2.7 (range: 2-20) weeks. The duration of benefit for blepharospasm, hemifacial spasm and cervical dystonia is shown in Table 2. In 13 patients the duration of benefit was unknown. The commonest complications are shown in Table 3. Adverse effects were commoner in female patients compared to males (30 versus 8; p < 0.001). The most frequently injected muscles in blepharospasm were the orbicularis oculi (n = 7). The orbicularis oculi and zygomaticus muscles were most often injected in hemifacial spasm (n = 17). The sternocleidomastoid, splenius capitis and semispinalis capitis (n = 12) were the most frequently injected in cervical dystonia. The results showed that there was significant difference in dosage increments between the two visits (75.5 ± 34 versus 82.0 ± 31; p < 0.001). In addition, the outcomes of patients with cervical dystonia were assessed pre-treatment and post-treatment using the TWSTRS scores. Our results revealed that there was significant improvement in TWSTRS scores post-treatment; that is improvement of cervical dystonia symptoms (12.5 7 – 18) versus 6 2.25 -10.75; p < 0.001). We attempted to identify potential outcome predictors. There was a great disparity between the number of consultants and registrars (21 30% consultants versus 49 70% registrars) injecting botulinum A toxin. There was however no difference between consultants and registrars on the outcome of treatment (p = 0.8) in terms of response to botulinum A toxin (good, partial or no response). DISCUSSION There have been no previous studies published in Africa regarding the safety and efficacy of botulinum toxin A in the treatment of the main focal dystonias affecting the head and neck. This study found cervical dystonia to be the commonest focal dystonia encountered at the botulinum toxin clinic. In our study, a median dose of 20 units, 25 units and 82.5 units for hemifacial spasm, blepharospasm and cervical dystonia respectively were required to elicit an optimal response. A retrospective chart review found that average doses of 14,5 units, 49,6 units and 137 units for hemifacial spasm, blepharospasm and cervical dystonia respectively, were required for an optimal response. 8 Another report found that doses of 25 units per eye for blepharospasm and 13,5 units for hemifacial produced responsiveness in 72% and 98,6% of patients respectively. 7 The doses observed in this study reflect individual variability in requirements and response to injection. The standard practice at our clinic is dose titration to achieve a maximum benefit while limiting adverse effects. In our study, 35 (50%) patients showed a good response, 22 (31.4%) showed a partial response and 4(5.7%) no response. A retrospective chart review found a good response to botulinum toxin injections in patients with hemifacial spasm (81.7%), with a moderate response in patients with blepharospasm and cervical dystonia, which is consistent with our findings. 8 In our study 5 (6,2%) patients did not respond to botulinum toxin injections. Lack of response to botulinum toxin for dystonia may occur for myriad reasons including immunoresistance due to the development of neutralising antibodies. 10 Our results indicate that there is a wide variation in the duration of relief of symptoms amongst our patients. Reports from other studies found the duration of relief to be between 12 and 16 weeks.8,13 Explanations for a short duration of relief of symptoms or clinical unresponsiveness include inappropriate muscle or injection site selection, underlying muscle changes such as the development of contractures, muscle atrophy or the formation of neutralising antibodies. 4,5Although not applicable to our study, a shorter duration of benefit can translate into more frequent visits to the botulinum toxin clinic resulting in multiple injections, inconvenience and higher travel expenditure as well as general patient discomfort and absenteeism from work. A high frequency of adverse effects was observed in our study. We found ptosis to be the commonest side effect in hemifacial spasm and blepharospasm, a finding consistent with most reports 5,7,8,12 Neck pain is a frequent and early feature of cervical dystonia as well as a common side effect following botulinum toxin injections.5 In our study neck pain was recorded as the commonest side effect following injections. Other commonly reported side effects in cervical dystonia include dysphagia, dry mouth and neck weakness.1,4 The muscle selection was based upon analysis of individual dystonic muscle involvement. The most frequent muscles injected in our series are consistent with other reports. 5,8 In patients with cervical dystonia we analysed the TWSTRS pre-treatment and post -treatment scores as an outcome measure to quantify any changes in the disability of patients from baseline to follow-up. This study showed a significant improvement in the scores pre-treatment and post-treatment, consistent with other reports. 6,10,11 We were unable to quantify changes in disability in patients with blepharospasm and hemifacial spasm due to unavailability of data. The majority of our injectors had between 1 and 5 years of experience and a small number had less than one year in treating dystonic patients. There was no difference between consultants and registrars with regards to the response (good, partial or no response) to treatment (p = 0.8). Explanations for the above may include a small study sample. There is a general consensus among experts that selection of the appropriate muscle and subsequent injection of the optimal dose are the most important determinants of the outcome of botulinum toxin treatment. 3Although the majority of our injectors had an adequate level of experience, the rate of complications were high, probably due to poor injection technique. There is a paucity of data in the literature with regards to the relationship between injector experience and the rate of complications. The main limitation is that the study is a hospital based retrospective chart review. A second limitation was the small sample size. Patients were identified using a booking book. This process might have led to patients been missed. Many patients’ files had no primary diagnosis at initial visit and specific adverse effects were not always recorded. A lack of data or of reliable data limited the scope of the analysis. CONCLUSION Botulinum toxin A was found to be safe and effective in the majority of patients with the main focal dystonias affecting the head and neck with cervical dystonia being the commonest focal dystonia at our clinic. These main focal dystonias were commoner in an older age group with a female preponderance, was not operator dependent and a well-known pattern of side effects was found. Acknowledgements I would like to acknowledge the support provided by my supervisor Dr Vinod Patel during this research project. Competing interests The authors declare that they have no financial or personal relationships, which may have inappropriately influenced them in writing this article. Authors’ contributions S.B (Sudika Bhola) and V B. Patel (Vinod Patel) conceptualized the study. S.B collected the data and drafted the article. 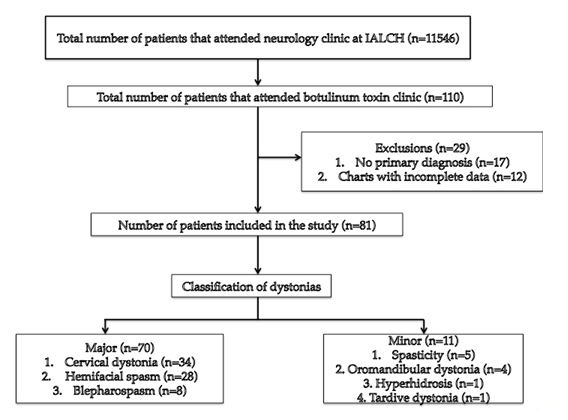 Figure 1 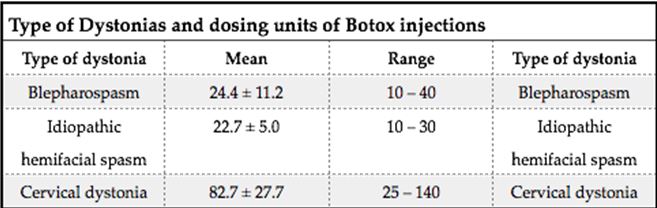 Table 1 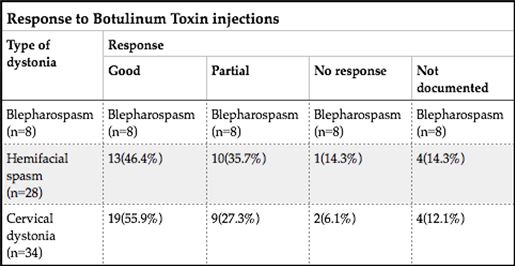 Table 2 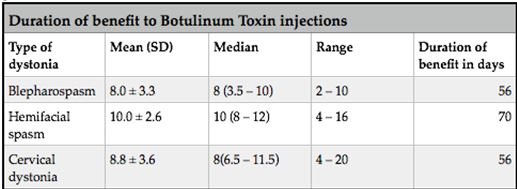 Table 3 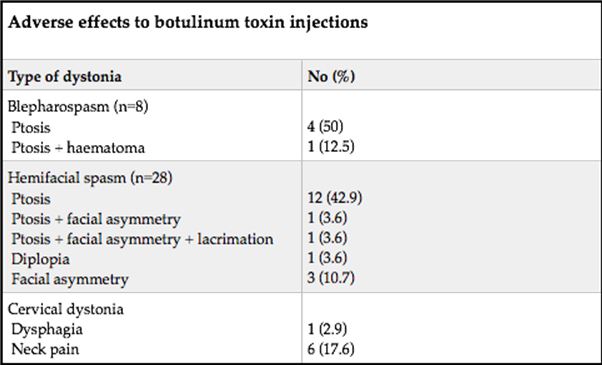 Table 4 REFERENCES
|
© 2002-2018 African Journal of Neurological Sciences.
All rights reserved. Terms of use.
Tous droits réservés. Termes d'Utilisation.
ISSN: 1992-2647