
|
|
|
CASE REPORT / CAS CLINIQUE
CAUDA EQUINA SYNDROME SECONDARY TO A POSTERIOR MIGRATION OF A DISC FRAGMENT
SYNDROME DE LA QUEUE DE CHEVAL SECONDAIRE A UNE MIGRATION POSTERIEURE D'UN FRAGMENT D'HERNIE DISCALE
E-Mail Contact - HILMANI Saïd :
hilmani.said@caramail.com
ABSTRACT Because of anatomical obstacls, migration of a disc fragment around the dural sac to the posterior epidural space is a rare phenomenon. Patients with this problem present generally with radiculopathy but may also present with cauda equina syndrom. We report a case of a 70 years old man who was complaining of a cauda equina syndrom secondary to a posterior disc fragment witch was diagnosed by MRI as a spondylodisitis, and confirmed later by the histo pathological examination. The patient was operated with laminectomy and total excision of the lesion. At the follow up examination the patient regained full neurological function 12 months post operatively. Key words: Cauda equina syndrome, MRI, Posterior disc herniation. RESUME Compte tenu de la considération anatomique, la migration d’un fragment discal autour du sac dural en particulier au niveau de l’espace postérieur est rare. La plupart des patients victimes de cette compression du fourreau dural se présentent dans un tableau de syndrome de la queue de cheval. Nous rapportons le cas d’un patient de 70 ans dont le diagnostic avait été suggéré par l’IRM et confirmé par la chirurgie et l’examen anatomo-pathologique. L’évolution a été marquée à un an, par une récupération complète du déficit neurologique. Mots clés : hernie discale, IRM, syndrome de la queue de cheval. INTRODUCTION Sequestered fragments are known to migrate in superior ; inferior and lateral directions within the spinal canal. Posterior epidural migration is very exceptional (3,6). The most frequent clinical presentation is radiculopathy, the cauda equina syndrom is rare. CASE REPORT A 70 years old man, was complaining of low back pains for one year after a fall, these pains failed to respond to medical therapie. Since 20 days, he experienced a progressif bilateral weakness of his lower limbs with bowel and bladder retention. The clinical examination revealed a flaccid paraplegia and a saddle anesthesia without other sensitif disturbances. Dorsolumbar radiographs and computed tomography scan showed important and extended degeneratif changes. MRI revealed an epidural process compressing the dural cord dorsally in the segment L1-L2. Tha process was hypointense on both T1 (fig1) and T2 (fig2), after gadolinium injection the lesion showed a ring-like enhancement (fig3). This process was believed to be epidural inflammation secondary to tumor or to spondylodisitis. In an emergency operation, bilateral complete laminectomy L1-L2 was performed with total process excision. The extirpated tissue had a yellowish appearance and a fibrotic consistency,with attachement to the dura, but without any communication with the intervertebral disc space anteriorly. The diagnosis of posteriorly migrated lumbar intervertebral disc was later confirmed histologically. Immediately, the patient had recupered 75% of his motor function. At the follow up examination, the patient regained full neurological function with a good bladder continence 12 months post operatively. DISCUSSION After posterior longitudinal ligament (PLL) rupture, lumbar intervertebral disc fragment most often migrates in the anterior epidural space, in rostral; caudal; and lateral directions. But posterior epidural migration is a very rare event, because of anatomical structures presented by the peridural membrane; sagittal midline septum; midline and dural ligament of hoffman; the PLL; the dura; epidural vessels; fat and the nerve root itself. A problem with any one of these barriers may facilitate posterior migration of disc fragments witch may be greater if the angle formed by the nerve root and the dural sac is obtuse at the site where the root exits (2,4). Heavy labor, skeletal traction, hypermobility and degenerative disc desease may have a principal rule in this migration (4,5). Our patient had in fact a very important arthrosis of the lumbar spine. Clinical presentation of a posterior migrated fragment is usually a severe radiculopathy (1) due to root compression, but it may also cause cauda equina syndrome. In the fewer cases of cauda equina syndrome secoandary to posterior disc fragment, reported in the published literature (1,2,5), the livel was L2-L3 or L3-L4, but In our patient this level was L1-L2, witch is the first case to our knowledge. MRI is the best technique to evaluate the cauda compression, but it is enable to make discrimination between sequestred disc materiel and intraspinal tumors or epidural inflammation secondary to a spondylodisitis (5), as primarly evoked in our patient. Sequestred fragment is usually hypointense on both T1 and T2 weighted images similar to disc herniation in other regions. But the difference is a ringlike contrast enhancement after injection of gadolinium in posterior migrating sequestre, witch indicates the presence of an inflammatory reponse with neovascularisation around the extruded tissue. This explained attachement to the dura so as seen in our patient. A definite diagnosis is difficult to make because a disc fragment in a posterior location has caracteristics similar to other common epidural lesions (4) as neoplasia; infection; hematoma. CONCLUSION Migration of sequestred disc fragment is a rare phenomenon. MRI confirme the posterior compression of the dural sac, but the differential diagnosis of sequestred fragment and other more common epidural lesions is not usually easy. After early diagnosis and surgery, the results are encouraging. 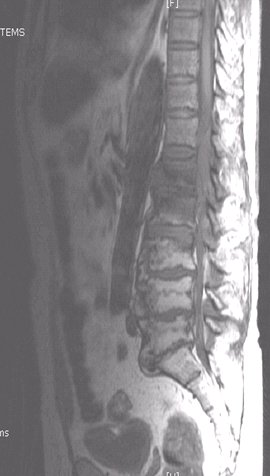 Figure 1 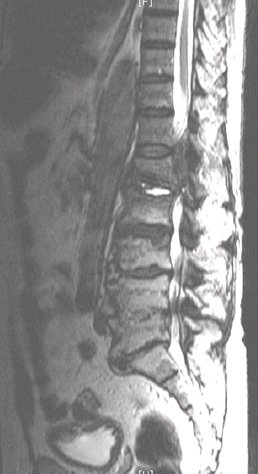 Figure 2 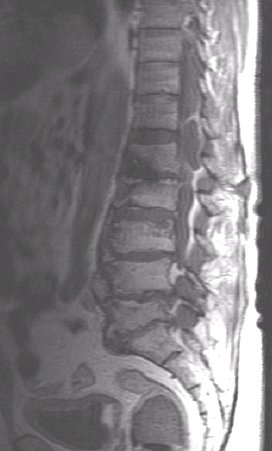 Figure 3 REFERENCES
|
© 2002-2018 African Journal of Neurological Sciences.
All rights reserved. Terms of use.
Tous droits réservés. Termes d'Utilisation.
ISSN: 1992-2647