
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
CLINICAL STUDIES / ETUDES CLINIQUES
CHRONIC SUBDURAL HAEMATOMA: CLINICAL PRESENTATION, SURGICAL TREATMENT AND OUTCOME AT THE LAGOS UNIVERSITY TEACHING HOSPITAL
HEMATOME SOUS-DURAL CHRONIQUE : PRESENTATION CLINIQUE, TRAITEMENT CHIRURGICAL ET RESULTATS AU CENTRE HOSPITALIER ET UNIVERSITAIRE LAGOS
E-Mail Contact - BANKOLE Olufemi Babatola :
f_baba@yahoo.com
ABSTRACT Background Objective Methods Results Conclusion Keywords: Chronic subdural haematoma, Nigeria, surgical outcome, treatment RESUME Introduction Objectif Méthode Results L’évacuation par trous de trepan, sous anesthésie générale a été la technique la fréquemment utilisée (96 %). Cinq patients ont eu en post opératoire des complications avec un résultat favorable dans 91,3 % des cas. Les taux de récidive et mortalité étaient respectivement de 12,5 % de 6,3 %. Conclusion Mots clés: Hématome sous-dural chronique, Nigéria, traitement chirurgical INTRODUCTION Chronic subdural hematoma (CSDH) is a relatively common problem in neurosurgical practice worldwide with favourable prognosis when treated adequately. The incidence is about 5 per 100,000 per year in the general population and is higher in the elderly rising to about 58 per 100,000 per year (17). The proportion of people aged 65 years and older is expected to double worldwide between 2000 and 2030, and a correspondingly large increase in incidence of CSDH is expected (10). A steady increase in the incidence has been observed in Nigeria because of increasing availability of computerized tomographic (CT) scan machines (1) and an apparent rise in life expectancy. Diagnosis of CSDH is still frequently missed or delayed in our environment with many patients often admitted and managed for other medical conditions before finally being referred on account of brain CT scan findings. In the western world anticoagulant or antiplatelet therapy, alcohol abuse and trauma to the head are frequent aetiologic factors for chronic subdural haematoma (8, 17, 20, 23,). The role of such factors except for trauma has not been documented in our environment. Pre-existing co-morbidities like myocardial and renal problems have also been reported as a significant cause of death in patients with chronic subdural hematoma. While surgical evacuation usually results in great improvement in neurological status and good outcome, still morbidity and mortality continues to occur especially in the elderly (3, 14). The purpose of this article is to present our experience of the surgical management of 73 patients with CSDH by evaluating their clinical features including predisposing factors, CT scan findings, surgical results and the postoperative outcome. MATERIALS AND METHODS Data were collected in two parts, the first part consisted of a retrospective review of cases of chronic subdural haematoma (CSDH) managed between May 2005 and June 2009 and the later part was a prospective review of cases from July 2009 till September 2010. A total of 73 patients CSDH were managed during the period under review, forty-nine patients had complete records, while twenty-four had incomplete medical records. Information extracted from records included the patient’s biodata, clinical features, aetiological factors, initial diagnosis and treatment before brain CT scan and subsequent referral to neurosurgery. For patients with history of trauma, the type and duration of trauma before presentation were documented. In all cases, diagnosis was based on brain CT scan or MRI findings which were correlated to the clinical presentation of the patients. The neurological status on admission by the neurosurgical unit was graded using the Glasgow Coma Scale (19) preoperatively and was reassessed in the first postoperative day. Management outcome was assessed using the Glasgow Outcome Score (GOS) (9) at discharge and during out-patient follow up. Based on GOS, patients were further grouped into two outcome categories; favourable (GOS: good recovery, moderate disability) and unfavourable (GOS: severe disability, persistent vegetative state and death) CSDH recurrence was defined as recurrence of neurologic symptoms and signs with demonstrable subdural collection on the operated side and compression of the brain surface observed in a repeat CT scan. RESULTS A total of 73 patients underwent 79 operations during the study period. The age range was 24 to 82 years, with majority of the patients between 61 – 70 years. (Table 1). A history of trauma was elucidated in thirty patients (63.5%); road traffic accident (56.7%) was the commonest cause of trauma, while falls accounted for 26. 7%. Among patients with history of trauma, 13 patients (43.3%) presented between 3 – 6 weeks of trauma, 9 patients (30%) presented after six weeks and 8 patients (26%) presented before 3 weeks (Table 2). None of the patients had received neurosurgical treatment for their reported head injury. Hypertension (22.9%) was the commonest co-morbidity; two patients had more than one co-morbid condition. Thirteen patients (43.3%) had history of significant alcohol use. Only 2 patients were on anticoagulant therapy prior to presentation. Eighty-nine percent of patients presented with headache while 62.5% had motor deficit. Among the forty-nine patients with complete records, twenty-five (52%) patients presented to the neurosurgery unit with a GCS of 15 – 14 and in 6 (12.5%) patients the GCS was less than 8. The diagnosis was delayed or initially missed in 50%, with 12 (25%) of the patients being managed initially for strokes before a brain CT scan was requested(Table 3); other initial diagnosis included seizure disorders (8.3%) and psychiatric illness. There was no side predilection in occurrence of CSDH. Bilateral CSDH occurred in 22 patients. Figure 1 shows the CT characteristics of the lesion: it was hypodense in majority of cases (50%), isodense in 28% , mixed density in 17% and hyperdense in 5%. Seventy patients (95.9%) had burr-hole craniostomy and haematoma evacuation while 3 patients had craniotomy. None of the patients had deterioration of GCS postoperatively. Most operations (72 operations, 95%) were performed under general endotracheal anaesthesia, with few cases done with local scalp infiltration with 1% lidocaine with Adrenaline 1:200,000. The surgical procedure included burr-hole craniostomy and drainage of haematoma using a single burr-hole for unilateral CSDH and two burr-holes, one on either side, for bilateral cases as a routine. Three patients underwent craniotomy and evacuation of haematoma: one of these three patients had calcified CSDH while the indication for craniotomy in the other two patients was not stated in the case file. Evacuation of the Subdural haematoma was achieved with the aid of a silastic feeding tube size 8FR and repeated irrigation of the subdural space with physiological saline solution followed by placement of a closed-system drain usually for 2 days. Routine post operative CT scan was not done. Antibiotic therapy was continued until drain removal and patients were mobilized from first postoperative day. After the first day, postoperative GCS was 15 – 14 in 34 (94.4%) and 13 – 9 in 2 (5.6%) patients. Figure 2 shows the preoperative and postoperative GCS score. Five patients developed postoperative complications; 2 patients developed seizures, 1 patient each developed an intracerebral haematoma (ICH), significant pneumocephalus and urinary retention. Outcome was favourable in the majority (91.3%) of patients with a recurrence rate of 12.5% and mortality rate of 6.3%. (Table 4). DISCUSSION Chronic subdural haematoma is a frequent cause of neurosurgical referral in our environment as it is in other parts of the world (2). There has been an increasing trend in the number of cases treated over the past decade in our hospital. The majority of the patients (50. 7%) were above 61 years-old and 87% were male. This is similar to what has been reported in other centres around the world with majority of the patients being males in the 7th decade or above (20). As in other parts of the world, CSDH is a disease of elderly, however, a sizeable number of young people was found in this study. Trauma to the head (63%) and alcohol use (43%) were prominent aetiological factor in this study with only very few patients (4%) on anticoagulant therapy. This finding is unlike what is reported in some centres in the western world where therapy with anticoagulant or antiplatelet was present in up to 40% and alcohol abuse in 6% of patients (20, 23). Santarius (17) reported a rate of 21% and 29% of patients on anticoagulants and antiplatelets respectively. Other known risk factors for CSDH are seizures, cerebrospinal shunts and arachnoid cyst patients (5). The exact mechanism leading to chronic subdural haematoma is not clear. The existence of an acute subdural haematoma or subdural hygroma has been found as the predisposing factor for the development of a chronic subdural hematoma, especially in elderly patients. The clinical manifestations of CSDH are protean and an initial misdiagnosis was commonplace in the pre CT era. Headache (89%) was the commonest symptom, followed by motor deficits (63%) mainly hemiparesis in our centre. Other symptoms are altered/fluctuating levels of consciousness and inappropriate behaviour/speech. Twenty-five percent of the patients were initially diagnosed and managed for stroke before neurosurgical referral after brain CT scan finding suggestive of a CSDH. Other initial diagnoses were seizure disorders, dementia and unexplained headache. In patients being managed for ¡®stroke’, there was significant delay before neurosurgical intervention due to a local practice of not routinely obtaining brain CT scans in stroke patients. It is necessary for physicians to have a high index of suspicion when treating such suspected stroke patients and make use of Brain CT scans which is diagnostic and is now readily available in many centres in Nigeria. The most common CT finding in CSDH is the hypodense, crescenteric extra axial lesion: this picture was seen in 54% of our patients. At other times, the haematoma was isodense (25%), with mixed density (13%), and much less commonly hyperdense (8%). This finding is consistent with reports from other centres, however a predominant finding of mixed density lesions has also been reported (1, 17). The cause of a hyperdense haematoma is the continuous rebleeding and the formation of membranes in the cavity. Magnetic resonance imaging (MRI) is useful to demarcate the various phases of the subdural haematoma and provides detailed information of the dimension, age and complexity of the haematoma. However, due to limited availability and increased cost, MRI is mainly indicated for the differential diagnosis of isodense or bilateral haematomas (21). There was no side predilection in the occurrence of CSDH and the lesion was bilateral in 32% of patients. Three of the six patients with recurrent CSDH had bilateral lesions initially. One patient with unilateral lesion and recurrence had additional risk factors (alcohol abuse and abnormal coagulation studies). This same patient developed postoperative intracerebral haematoma with resultant dysphasia, which resolved with conservative treatment. The recurrence rate of 12.5% found in this study is similar to rates of 9.8% and 14.3% among patients treated with burr hole craniostomy and drainage in other centres (13,17,22 23) and lower than 18% and 33% amongst patients treated with no drain (17, 23). These results are consistent with a positive effect of drains in prevention of postoperative recurrent collections, and their use could avoid repeated operations and additional time in hospital. The use of drain has not been found to be associated with increased surgical complications (4, 17, 16, 23). A complication rate of 6.8% (five patients) is comparable to reports from other centers (15, 17). CONCLUSION In our environment, there is a noticeable increase in patients presenting with chronic subdural haematoma. A high index of suspicion will improve the timeliness of diagnosis and therapy. Despite frequent delay in making diagnosis and institution of treatment, surgery in form of burrhole craniostomy and closed-system drainage is associated with favourable outcome in the majority of patients, many of whom are elderly. TABLES Table 1: Demographic characteristics of patients
Table 2: Risk factors for CSDH
Table 3: Clinical presentation of CSDH
Table 4: Management and Outcome
FIGURES  Figure 1 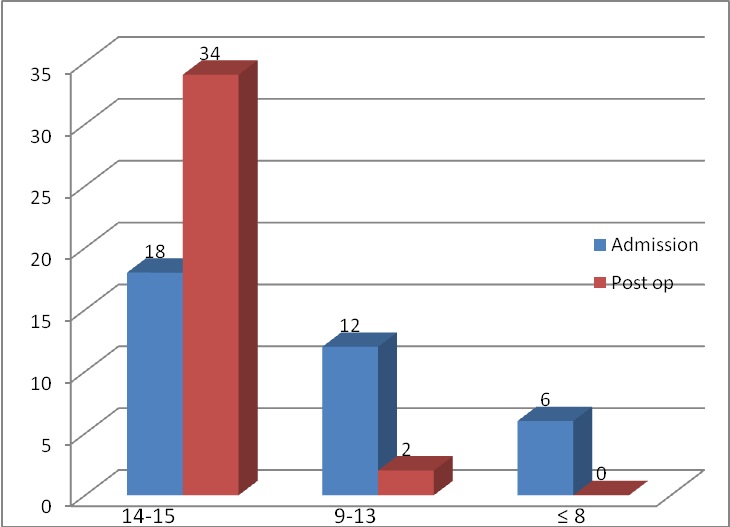 Figure 2 REFERENCES
|
© 2002-2018 African Journal of Neurological Sciences.
All rights reserved. Terms of use.
Tous droits réservés. Termes d'Utilisation.
ISSN: 1992-2647