
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
ORIGINAL PAPERS / ARTICLES ORIGINAUX
EVALUATION AND MODIFICATION OF KANG’S MRI METHOD OF GRADING CERVICAL SPINAL CANAL STENOSIS AMONG AFRICAN PATIENTS: AN INITIAL STUDY.
ÉVALUATION ET MODIFICATION DE LA METHODE D'EVALUATION MRI DE KANG DANS LA STENOSE CANALAIRE CERVICALE CHEZ LES PATIENTS AFRICAINS : ETUDE PRELIMINAIRE
E-Mail Contact - OLARINOYE-AKOREDE Sefiya Adebanke :
olarinoyebs@yahoo.com
ABSTRACT Background Objective Materials and methods Results Keywords: Cervical Stenosis, Kang, MRI INTRODUCTION The size of the spinal canal and integrity of its content (mainly the cord) are the key prognosticating parameters that are considered when evaluating a patient for spinal disease. Cord compression could be developmental or degenerative and in adults, the commonest cause of cervical myelopathy is spondylosis.1, 2 The smaller diameter of the cervical canal leads to greater symptoms for relatively smaller degrees of stenosis than its lumbar equivalent. Earlier reports which attempted to assess cervical canal stenosis, started with cadarveric and radiographic studies.3-8 Plain radiographs are largely obsolete now because of inherent radiation risks, magnification errors and lack of soft tissue detail although CT it is still useful for osseous details. MRI on the other hand is the choice modality in assessing the spine, overcoming the radiographic limitations. Even with this superior modality, its use in diagnosing patients with cervical myelopathy has not been fully harmonized worldwide. Descriptive terminologies such as mild, moderate, severe stenosis 2, 9, 10, 11 are subjective, and vary in definition between different interpreters. Nagata et al12 and Harrop et al13 focused on the spinal cord but not the spinal canal while Muhle13 and Larsson11 considered the cord and canal but were silent about T2W signal intensity (SI) of the cord, which itself is a strong prognosticating factor for patients with cervical spondylotic myelopathy (CSM).1, 14-17 The Kang method was developed based on the preexisting method of Muhle, so as to allow for assessment of the canal, spinal cord and cord parenchymal changes. In Nigerian literature, we are not aware of any method of grading stenosis either using radiographic methods or MRI. This study to the best of our knowledge is the first MRI documentation on grading of cervical stenosis in our environment. It aimed to assess the reliability and adaptability of a modified Kang method in our practice; and to build a data base for future research. MATERIALS AND METHODS Case selection: Approval for this study was given by the hospital’s ethics committee. A total number of 140 patients were referred for cervical MRI scan during the two year period of this study. Out of these, MRI scans of 52 patients with cervical spondylosis were examined for this review. Our basis for exclusion included age ie < 16 years, post traumatic/surgery, spinal tumor/infection, and poor quality image. Our study population comprised of 36 males and 16 females. Their bio data and clinical history were obtained from patients' hospital records. Image acquisition Only one MRI machine (0.2Tesla MAGNETOM Concerto Siemens Medical) was used to image all patients. T1 and T2weighted images were acquired using the fast spin echo technique. Patients were imaged lying supine with the head in neutral position and the use of a neck coil. The imaging protocols were as follows:
Image analysis 2 experienced radiologists OAS and IPO with 8 years and 12 years’ experience respectively were presented with a schematic representation of the grading method (fig.1).10 The examiners were blind to the patients’ clinical information and MRI reports. Each examiner graded all 52 images at 4 cervical levels ie C3/4, 4/5, 5/6 and 6/7. Mid sagittal as well as immediate adjacent parasagittal images were studied in order to allow for grading to be done at the point of maximum compression. The examiners employed the method proposed by Kang (Fig.2) however grades were modified and classified as follows:
Statistical analysis Statistical analysis was done by SPSS version 20 (Chicago Illinois). Percentage inter-observer agreement among the 2 Radiologists was calculated using the kappa statistics. This was also calculated for sub categories among the 4 grades: for distinguishing between presence or absence of stenosis (grade 0 vs 1,2,3), between significant and non-significant stenosis (0,1 vs 2,3), and for presence or absence of cord injury ie T2W signal change within the cord (grades 0,1,2 vs grade 3). The agreement was then rated according to Landis and Koch19,. as follow: kappa values of 0-0.2 indicated slight agreement, 0.21-0.4 indicated fair agreement, 0.41-0.60 indicated moderate agreement, 0.61-0.8 indicated substantial agreement, and 0.81 or greater indicated excellent agreement RESULTS In this study, 52 patients were examined independently and they were made up of 36 (69.2%) males, 16 (30.7%) females. Age range was 36 years to 78 years old (mean age 56.1years SD 10.87years). For both examiners, grade 0 was the most prevalent grade scored as most patients had insignificant stenosis (grade 0, 1) Fig 6. The frequency of stenosis by cervical level is shown in Table 1. C6/7 level was least compressed (grade 0= 70%) while the level more severely affected (grade 3) was C3/4 followed by C5/6. The Inter observer agreement and kappa statistics for various distinctions in each level is shown in Table 2. The overall inter-observer agreement was high (k= 0.65 to 0.87). For most levels, the agreement was substantial to almost perfect except at the lowest cervical level. The distinction between the presence of stenosis, how significant, and the presence of cord injury was almost perfect at C3/4 levels (> 90%; k= 0.81 – 0.85), substantial to almost perfect at C4/5 (86.5-92.6%; k= 0.65 – 0.85) and C5/C6 (88.5-98.1%; k= 0.79-0.87 Table 3 shows analysis of symptoms with respect to distinction between the grades. Correlation was made between significance of stenosis ie grades 0, 1(insignificant) and 2, 3 (significant) versus neurologic symptoms. 22 (84.6%) of the 26 patients with significant stenosis had neurologic symptoms while 15(57.6%) of patients with non-significant stenosis did not have neurologic symptoms. DISCUSSION The need to standardize methods of assessing cervical stenosis has led to recent evolutionary studies. Larsson et al11 defined mild, moderate and severe stenosis to mean subarachnoid space obliteration less than 50%, greater than 50% and compression of the cord respectively while Muhle at al13 used a 4 point grading system where grade 0 meant normal, grade1 was partial obliteration, grade 2 was complete obliteration of anterior or posterior subarachnoid space, and grade 3 meant cord displacement or compression. Kang et al modified the Muhle classification in order to incorporate SI T2Weighted cord changes. The reliability of Kang method was assessed by Park et al20 while our presentation examined and modified the Kang study, with review of previous literature. We modified the original Kang grade 1 by subdividing into grades1a and 1b as follows:
Results from this study showed a significant level of agreement which is higher than the agreement in the original Kang study which ranged from 63%-64%. The improvement in the agreements is largely due to the fact that the observers in our study had both schematic and pictorial representations of the grading system before the patients were scored although they were blind to clinical information and MRI reports. In a study by Stafirra et al2 using CT and MRI in which they had no instructions or guidelines, there was significant disagreement among interpreters. Inter-observer agreement for level, degree and cause for CT myelogram was k= 0.5, 0.26, 0.32; for MRI k =0.6, 0.31, 0.22 Our study therefore supports the fact that the modified Kang system can be taught and be understood by Radiologists and managing Physicians. The prevalence of each grade did not differ significantly between the 2 readers in our study. We also assessed clinical value offered by the grading system by sub categorizing the grades. Grades 0 vs. 1, 2, 3 (absence vs. presence of stenosis), % agreement was almost perfect across C3-6 (>80%, k between 0.65 – 0.85) but moderate at C6/7 (80.8%, k=0.57). For grades 0, 1 vs. 2, 3 (non-significant vs. significant stenosis) % agreement was greater than 90% with k values of 0.81, 0.85, 0.87 respectively at C3-6 levels; and 0.38 at C6/7. While for 0, 1, 2 vs 3(presence of SI within the cord, the % agreement and k values were 98.1%, k=0.65, 92%, and 98.1%, k=0.79 and 98.1%, k=0.00 across C3/4 to C6/7 levels. These results show that agreement between the examiners were generally better at the C3-6 than at C6/7. The study by Kang only assessed 3 cervical levels and did not examine C6/7. Our results showed better agreements than Kang’s which recorded 81-85% ((0, 1 vs2, 3) and 91-95% (0, 1, 2 vs. 3). In the study by Park et al, they studied 4 cervical levels as we did but they had a slightly different modification from ours. In addition, they correlated their findings with clinical symptoms and neurologic examination. They however also recorded a higher prevalence of grade 0 similar to our report, unlike Kang who reported grade 1 to be the most prevalent grade. Again, this could be explained by the fact that in using older subjects (>60 years), Kang study would have fewer normal findings since spondylosis is mostly a process of aging. We also recorded that the cervical level least prone to compression was C6/7, which had the highest prevalence of grade 0. When we considered patients’ symptoms, most (22 out of the 26) patients with significant stenosis (grades 2 and 3) also had positive neurologic symptoms. All (100%) of patients with spinal cord injury, signified by high T2 signal intensity were found to have severe symptoms. However, patients’ symptoms compared less well with grades 0 and 1(insignificant stenosis). 42.3% of patients with non-significant stenosis had positive symptoms. But being a retrospective study, the severity of symptoms could not be assessed clinically. Our findings showed that out of 19 patients who were asymptomatic, 4 had severe stenosis and therefore we entertained the possibility of asymptomatic congenital stenosis. Our modification of Kang grade 1 into 1a and 1b did not lead to an upgrade or downgrade from the overall grading system and will not significantly affect the patients’ management in the short term. However, it conveys a relevant subtle clinical message of early onset stenosis. We believe that in our environment, the modified Kang will allow for surveillance of individuals who had mild stenosis and would otherwise be categorized as no stenosis (i.e grade 0) in the original Kang system. This is especially since we operate on a low field MRI machines in most parts of the country, and also because our patients were mostly much younger (less than 60 years), compared with the Kang study and with time their spondylotic process would increase as they get older. Patients with mild stenosis have been found to progress over time in previous reports. 21,22 CONCLUSION Our study is an initial preliminary retrospective study, a revised Kang scoring system (2016) and represents the first documentation in Nigeria. We agree that it is a method that can be taught and understood; and is reproducible on even low Tesla MR Scanners. Limitations The low field strength (0.2Tesla) scanner used may have affected image quality especially in axial plane (higher strengths were not available in our locality). Hence the use of only sagittal images where the delineation of the subarachnoid space and the spinal cord was clearer.
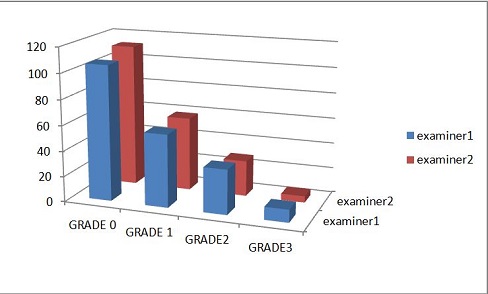 Figure 6 Table 1. The frequency of stenosis by cervical level
Table.2 The Interobserver agreement and kappa statistics for various sub categories of grades
Table. 3 Analysis of symptoms with respect to distinction between mild stenosis( ie grades 0 &1) and severe stenosis (grades 2 & 3)
REFERENCES
|
© 2002-2018 African Journal of Neurological Sciences.
All rights reserved. Terms of use.
Tous droits réservés. Termes d'Utilisation.
ISSN: 1992-2647


