CLINICAL STUDIES / ETUDES CLINIQUES
SURGICAL TREATMENT AND OUTCOME OF 195 CASES OF NON ACUTE SUBDURAL HAEMATOMA AT THE YAOUNDÉ CENTRAL HOSPITAL: THE NEED FOR LANDMARKED BURR HOLES
TRAITEMENT CHIRURGICAL ET DEVENIR DE 195 CAS D'HEMATOMES SOUS-DURAUX NON AIGUS A L'HOPITAL CENTRAL DE YAOUNDE: NECESSITE D'UN POINT DE REPERE POUR LE TROU DE TREPAN
- Service de Neurochirurgie, Hôpital Central de Yaoundé
- Anaesthesiology and Reanimation Department, Yaoundé Central Hospital, Cameroon
- Neurology Department, Douala General Hospital, Douala, Cameroon
- Department of Anaesthesiology and Reanimation, Yaoundé Gynaeco-Obstetrics and Paediatrics Hospital, Yaoundé, Cameroon
ABSTRACT
Background
Chronic subdural hematoma (CSDH) is frequent neurosurgical pathology but many issues related to the surgical procedure are still discussed.
Objective
To present our experience in the treatment of Non Acute Subdural Hematoma (NASH).
Methodology
This retrospective study included patients operated for NASH at the Yaoundé Central Hospital from January 2000 to September 2008. Surgery consisted of one burr hole (92.4%), 2 burr holes (3.6%), trephine or craniotomy (4%). All patients underwent per operative irrigation, a 48 hour-postoperative drainage and supine position. The burr hole was performed at the thickest point of the hematoma which in most cases corresponded to the intersection between the superior temporal line and the coronal suture.
Results
One hundred and ninety five consecutive patients were included: 155 males and 40 females. The mean age was 55 years. The past medical history revealed head injury (81.5%), alcoholism (12.9%), epilepsy (4.1%) or anticoagulation therapy (1.5%). The collection was unilateral (72.8%), bilateral (26.7%) or interhemispheric (1 case). On CT scan, the lesion was hypodense (79.9%), isodense (17.4%) or mixed (2.7%). The mortality rate was 2.5% while the recurrence rate was 3.7% after a 6 to 9 months follow-up period. The main recurrence factor was the inappropriate location of the burr hole.
Conclusion
One burr hole drainage is an effective and safe method for the treatment of NASH. The location of the burr hole is an important factor of recurrence. There is a need for Landmarked burr holes.
Keywords : Burr hole, Cameroon , Outcome, Chronic subdural hematoma, Treatment.
RESUME
Description
L’hématome sous-dural chronique (HSDC) est une pathologie neurochirurgicale fréquente dont certains aspects du traitement chirurgical restent débattus.
Objectif
Présenter notre expérience dans le traitement des Hématomes sous duraux non aigus (HSNA).
Méthodologie
Cette étude rétrospective incluait tous les patients opérés d’un Hématome Sous dural Non Aigu (HSNA) à l’hôpital Central de Yaoundé de Janvier 2000 à Septembre 2008. Le traitement chirurgical a consisté en la réalisation d’un trou de trépan (92,4%), de 2 trous de trépan (3,6%), d’une trépanation à la tréphine ou d’une craniotomie (4%). Tous les patients ont bénéficié d’une irrigation per opératoire, d’un drainage post opératoire de 48 heures (patient en décubitus). Le trou de trépan était réalisé au point d’épaisseur maximale de l’hématome qui correspondait dans la majorité des cas, à l’intersection entre la crête temporale supérieure et la suture coronale.
Résultats
Cent quatre quinze patients dont 155 sujets de sexe masculin et 40 de sexe féminin ont été inclus. L’âge moyen de notre échantillon était de 55 ans. Chez certains patients, nous avons retrouvé des facteurs de risqué tels: le traumatisme crânien (81,5%), l’alcoolisme (12,9%), l’épilepsie (4,1%) ou l’usage d’anticoagulants (1,5%). Au scanner cérébral, l’hématome était unilatéral (72,8%), bilatéral (26,7%) ou inter hémisphérique (1 cas). Il était par ailleurs hypo dense (79,9%), iso dense (17,4%) ou mixte (2,5%). Le taux de décès per opératoire était de 2,6% et le taux de récidive de 3,7% au terme d’un suivi de 6 à 9 mois. Le principal facteur de récidive était représenté par la localisation inappropriée du trou de trépan.
Conclusion
La réalisation d’un trou de trépan unique est une technique efficace pour le traitement HSNA. Sa localisation est un facteur important pour la survenue des récidives. Il y a une nécessité de définir des repères pour la localisation du trou de trépan.
Mots clés : trou de trépan, Cameroun, Hématome sous-dural chronique, Traitement.
INTRODUCTION
Chronic subdural hematoma (CSDH) is a frequent pathology in Neurosurgery. Burr hole craniostomy, twist drill craniostomy or craniotomy are the different options for surgical treatment. Burr hole craniostomy is the common technique. The recurrence rate after the treatment ranges from 3.4 to 30% according to the literature (1, 2, 3, 4, 5, 6, 7, 9, 11, 15, 16, 17, 20, 23, 24). Many reports attempted to identify risk factors associated with the recurrence of CSDH. The number of burr holes appeared in some studies as a major factor of recurrence (24).
Although the result of the surgical treatment of CSDH can be spectacular, this surgery is no longer a fascination for neurosurgeons that are more impressed by the surgery of aneurysms, skull base tumours or functional neurosurgery. Many cases of failure or recurrence may be related to poor technique, mistakes and carelessness during the procedure.
The authors present their experience with 195 cases of surgically treated Non Acute Subdural Hematoma (NASH) at the Neurosurgery Department of the Yaoundé Central Hospital, Cameroon.
PATIENTS AND METHODS
All adult patients operated for Non Acute Subdural Hematoma (NASH) at the Yaoundé Central Hospital from January 2000 to September 2008 were retrospectively included in this study. The Yaoundé Central Hospital is the most important university hospital in Yaoundé. All patients were operated by the first (5%) or the second senior surgeon (92%) of the team and rarely by residents. Local anaesthesia was common (96.3%).
Most of the time, surgery consisted of burr hole drainage. Other techniques (trephine and craniotomy) were rarely used. The indication of each technique depended on the surgeon’s personal experience.
The site of the burr hole was located at the thickest point of the haematoma which in most cases was near the intersection of the superior temporal (insertion of the temporal muscle) line and the coronal suture on the CT scan (fig2). The burr hole was done near the insertion of the temporal muscles which is a constant landmark, just before or after the coronal suture. The diameter of the hole was 1.6 cm. The subdural cavity was irrigated continuously with isotonic saline (250 to 500 cc) through a small subdural soft catheter which allowed constant evacuation all around until the fluid became clear. A drainage system without suction was connected to an extracranial subgaleal redon catheter (CH n° 16 with its holes in front of the burr hole) for 48 hours. The patients were kept (not strict) in supine position for 48 hours. The lower limbs were mobilized early and constantly to minimize the risk of deep venous thrombosis. Patients were rehydrated with intravenous isotonic saline.
Patients were followed up at 3 months, 6 and 9 months. The patients lost to control were contacted by phone. Post operative CT scan (fig4) was performed (14 cases) only when the symptoms (headaches, altered consciousness, focal signs or seizures) persisted or recurred (7 cases).
RESULTS
One hundred and ninety five adult patients operated for NASH were included: 155 males (79.5%) and 40 females (20.5%) were involved. The mean age was 55 years (range from 21 to 89 years). Head injuries (81.5%), chronic alcoholism (12.9%), epilepsy (4.1%) or anticoagulation therapy (1.5%) were found to be the aetiological factors associated. One case was iatrogenic (complication of surgery performed for another purpose). Various types of accident were described: road traffic accidents (124 cases), assaults (30 cases), falls from the patient height (20 cases), and falls from high floors (10 cases). In 11 cases, the type of accident was not specified.
The clinical presentation included headaches (189 cases), altered consciousness (100 cases), hemiparesis (138 cases), aphasia (7 cases), blindness (1 case) and sphincter dysfunction (1 case). The Glasgow Coma Score was 15 (95 cases), between 8 and 14 (71 cases) or below 8 (29 cases). Signs of temporal herniation (coma and unilateral mydriasis) were present in 10 cases.
The diagnosis was confirmed by CT scan in all cases. The collection was unilateral (79.9%), bilateral (26.7%) or interhemispheric (1 case). It appeared hypodense (79.9%), isodense (17.4%) or mixed (2.7%) including hypo- and hyperdense components. The delay between the accident and the surgical intervention was less than 15 days (10.5%) or ranged from 15 to 30 days (21%), 30 to 45 days (10.5%), 45 to 60 days (21.1%), 60 to 75 days (7%), 75 to 90 days (10,5%) or more than 90 days (19.4%).
The treatment consisted of one burr hole drainage (92.4%), 2 burr holes per hematoma (3.6%), trephine (2%) or craniotomy (2%). The anaesthesia was local (96.3%) or general (3.7%). General anaesthesia was indicated in patients with respiratory disorders (2.05%) or in the rare cases of craniotomy. A post operative drainage was kept for 48 hours. The patients stayed in supine position for 48 hours (not strict). The drain produced 100 to 500 cc of liquid.
Five patients died in the immediate post operative period (2.5%) due to pulmonary embolism (one patient), inhalation pneumonia in comatose patients with delayed management (3 cases) and brain herniation (one case). During the follow-up, 2 patients died of unrelated causes (cerebrovascular accident at 2 and 3 months). Wounds infections were observed in 2 cases. The sequellae were motor deficits (4 patients), cognitive disorders (3 patients) and epilepsy (2 cases).
Patients were followed up at 3 months (99%), 6 months (80%) and 9 months (75%). Feed back or data of patients lost to control were obtained by phone (from patients themselves or from relatives). Recurrence occurred in 6 patients (3.7%). The postoperative CT scan performed in those cases showed that the burr hole was located at the periphery of the haematoma.
DISCUSSION
Many procedures have been used in the treatment of chronic subdural haematomas (CSDH) with more or less success; but each technique failed to clearly demonstrate its superiority on the others: craniotomy or burr hole (5, 7, 10, 20, 22) , one or 2 burr holes (3, 17, 24), twist drill craniostomy or larger holes (4, 11, 16, 17, 23), irrigation, per or postoperative drainage (15, 16, 19, 25), post-operative 48-72 hours supine position. In many centres, there is no standard nor a consensual protocol of treatment adopted. The decision to perform one or the other technique is taken according to the personal experience or preference of the surgeon.
These procedures are based on 2 fundamental principles. The first principle is the evacuation of the hematoma and to empty the cavity during the intervention (twist drill craniostomy, burr hole, craniotomy, trephine or shunting of the subdural space) (3, 4, 5, 6, 7, 15, 16, 17, 25), and eventually in the post operative period (drains). The second principle is the replacement of the pathologic fluid (containing fibrin degradation products and plasminogen activator) by an absorbable fluid (ringer lactate, saline 9%, oxygen) through continuous irrigation-drainage of the subdural space (1, 2, 19). Other debated issues concern supine position and rehydration which are supposed to facilitate the first mechanism. The purpose in our institution was to combine the different mechanisms (burr hole evacuation, intra operative irrigation and postoperative drainage) to lower the recurrence rate and complications. We thought that patients might be protected from recurrence by one mechanism if the other ones failed. If the emptying of the cavity was not complete, the patient might be protected if the residual collection was absorbable (saline or extremely diluted haematoma) or subsequently evacuated during the post-operative period.
The location of the burr hole may be an important factor associated to the recurrence rate of NASH after surgery. The burr hole should be performed at the thickest point of the hematoma. When the burr hole is performed at the periphery of the hematoma, the hole might be covered by the re-expanding brain tissue so that the residual haematoma would be trapped; this would hamper the post operative subgaleal evacuation and resorption. In our experience, the thickest point of the hematoma was always near the intersection between the superior temporal line (insertion of the temporal muscle) and the coronal suture. This is probably due to the morphology of the cranium and secondly to the fixity of the brain which is attached on the sagittal midline and the temporal base by the bridging veins. In the antero-posterior plan the thickest site was just behind the coronal suture but the frontal and occipital extension varied from one case to another. Most of the hematomas were fronto-parietal. To avoid hazardous placement of burr hole, one should systematically identify the thickest point of the hematoma on CT scan, then correlate to the coronal suture and to the superior temporal line (the coronal suture and the insertion of the temporal muscle are easily recognized on CT scan and during the intervention) which constitute a constant landmark (figures 2). In many centres, surgery of subdural hematoma is performed by younger neurosurgeons or trainees with less experience. In many cases, it is performed without supervision. This highlights the importance of standard landmarks for the location of the burr holes. This may be an important factor in the outcome of patients with subdural haematoma. Further CT scan studies with measurements of the dimensions of the hematoma need to be carried out to ascertain this observation on the thickest point of the hematoma.
The number of burr holes has been considered as an independent predictive factor of recurrence and complications of CSDHs (24). In a previous study, the recurrence rate was reduced from 29% to 5% using 2 burr holes instead of one (24). The recurrence rate of CSDH ranges from 3.4 to 30% according to the literature (1, 2, 3, 4, 5, 6, 7, 9, 11, 15, 16, 17, 20, 23, 26). Many studies attempted to identify risk factors associated with the recurrence, but many aspects of the treatment are still debated. The recurrence rate in this series was only 3.7% using only one burr hole. Although our patients were younger with less co-morbidity than those reported in many occidental series, the recurrence was mostly associated to the inappropriate placement of the burr hole at the periphery of the hematoma. These cases were performed mostly by trainees.
Other methods (per operative irrigation-drainage, supine position, rigorous haemostasis on osteodural edges) used in this series may have contributed to the reduction of the recurrence rate. The tip of the drain should not be necessarily in the subdural space. In our series the size of the drain was large (Redon catheter n°16) and the tip was placed in the subgaleal space (figure 3) in front of the burr hole. This may minimise cortical injuries, CSF leakage and infections. The peroperative irrigation may play an important role in the treatment of NASH. The pathological solution is replaced by a physiologic absorbable solution (isotonic saline) (figure 4). The pathological solution contains anticoagulant proteins and other proteins responsible for recurrent haemorrhage (8, 12, 13, 14, 18, 26, 27, 28). This may be especially helpful if the brain fails to re-expand immediately. The second advantage of irrigation is to prevent air influx into the skull. The supine position may maintain an intracranial pressure which is high enough to facilitate the drainage of the residual collection in the subdural space.
CONCLUSION
One burr hole is an effective and safe method to evacuate Non Acute Subdural Hematoma (NASH). The number of holes does not appear as a factor of recurrence in this study. The appropriate location of the burr hole at the centre of the hematoma is important. This highlights the necessity of standard landmarks for making burr holes. The coronal suture and the temporal line are helpful landmarks. Other factors as intra-operative irrigation, the post-operative close system drainage, the postoperative supine position may have probably played an important role in the surgical outcome of our patients. The combination of these different issues during the same surgical procedure may reduce the recurrence rate and the incidence of post operative complications. Surgery of subdural haematomas should not be considered as the surgery of trainees without supervision.
| AUTHORS CONTRIBUTION |
| DVP designed the study and operated 95% of the patients. YTY recruited the patients. DVP and YTY analyzed the data and DVP wrote the article. All authors made critical contributions to the article. |
| ACKNOWLEGMENTS |
| The authors are grateful to all patients and to the staff of the neurology and neurosurgery departments at the Yaoundé Central Hospital, for their remarkable collaboration and support in this study. |
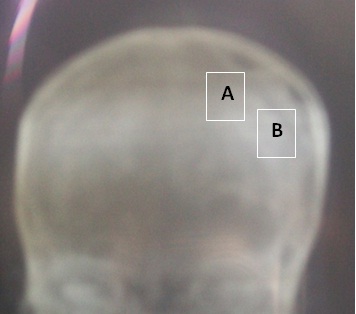
Figure1
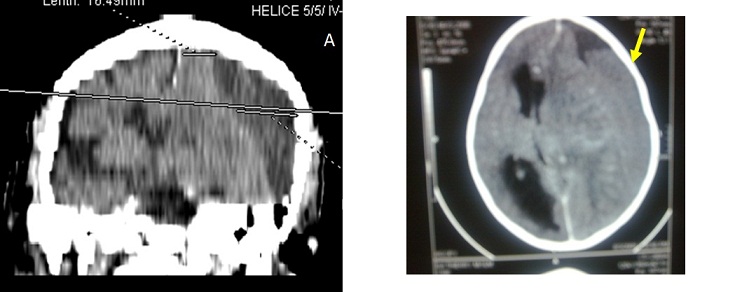
Figure 2
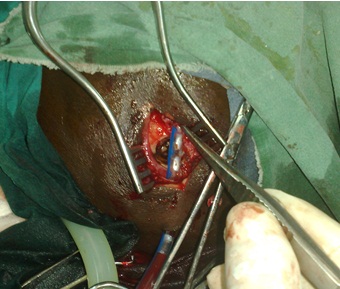
Figure 3
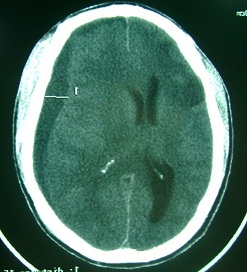
Figure 4
REFERENCES
- AOKI N. A new therapeutic method for chronic subdural hematoma in adults: replacement of the hematoma with oxygen via percutaneous subdural tapping. Surg Neurol. 1992;38:253-256.
- AOKI N, SAKAI T. Computed tomography features immediately after replacement of hematoma with oxygen through percutaneous subdural tapping for the treatment of chronic subdural haematoma in adults. Acta Neurochir Wien. 1993;120:44-46.
- BENZEL E, BRIDGES RT, HADDEN T, ORRISON W. The single burr hole technique for the evacuation of non-acute subdural haematoma. J Trauma. 1994;36(2):190-194.
- CAMEL M, GRUBB RL, Jr. Treatment of chronic subdural hematoma by-twist drill craniostomy with continuous catheter drainage. J Neurosurg. 1986;65:183-187.
- DAKURAH TK, IDDRISSU M, WEPEBA G, NUAMAH I. Chronic subdural haematoma ; review of 96 cases attending the Korle B Teaching Hospital, Accra. West Afr J Med. 2005;24(4 ):283-286.
- DESTANDAU J, DARTIGUES JF, COHADON F. Hématome sous-dural chronique de l’adulte : facteurs pronostiques de la chirurgie. A propos de 100 cas. Neurochir. 1987;33:17-22.
- DONGMO L, JUIMO AG, ELOUNDOU NJ, NJAMNSHI AK, AVDEEVA V, TIYOU C. Hématome sous-dural chronique au cameroun : aspects cliniques, radiologiques et thérapeutiques : à propos de 43 cas. Med Afr Noire. 1999;46(3):158-161.
- EIR B, GORDON P. Factors affecting coagulation: fibrinolysis in chronic subdural fluid collections. J Neurosurg. 1983;58:242-245.
- ERNESTUS RI, BELDZINSKI P, LANFERMANN H, KLUG N. Chronic subdural hematoma : surgical treatment and outcome in 104 patients. Surg Neurol. 1997;48:220-225.
- HAMILTON MG, FRIZZEL JB, TRANMER BI. Chronic subdural hematoma: the role for craniotomy reevaluated. Neurosurg. 1993;33:67-72.
- HUBSCHMANN OR. Twist drill craniostomy in the treatment of chronic and subacute subdural hematomas in severe and elderly patients. Neurosurg 1980; 6(3) : 233 – 236.
- ITO H, KOMAI T, YAMAMOTO S. Fibrinolytic enzymes in the lining wall of chronic subdural hematoma. J Neurosurg. 1978;48:197-200.
- ITO H, YAMAMOTO S, KOMAI T, MIZUKOSHI H. Role of local hyperfibrinolysis in the etiology of chronic subdural hematoma. J Neurosurg. 1976;45:26-31.
- LABADIE LE, GLOVER D. Local alteration of hemostatic fibrinolytic mechanisms in reforming subdural hematomas. Neurol. 1975;25:669-675.
- MARKWALDER TM, SEILER RW. Chronic subdural hematomas: to drain or not to drain? Neurosurg. 1985;16(2):185-188.
- MARKWALDER TM, STEINSIEPE KF, ROHNER M, REICHENBACH W, MARKWALDER H. The course of chronic subdural hematomas after burr hole craniostomy and closed-system drainage. J Neurosurg. 1981;55:390-396.
- OKADA Y, AKAI T, OKAMOTO K, IIDA T, TAKATA H, IIZUKA H. A comparative study of the treatment of chronic subdural haematoma: burr hole drainage versus burr hole irrigation. Surg Neurol. 2002;57(6):405-409.
- PFELBAUM RI, GUTHKELCH AN, SHULMAN K. Experimental production of chronic subdural hematomas. J Neurosurg. 1974;40:336-346.
- RAM Z, HADANI M, SAHAR A, SPIEGELMAN R. Continuous irrigation-drainage of the subdural space for the treatment of chronic subdural haematoma: a prospective clinical trial. Acta Neurochir Wien. 1993;120 :40-43.
- SAKHO Y, KABRE A, BADIANE S, BA M, GUEYE M. L’hématome sous-dural chronique de l’adulte au Sénégal. A propos de 118 cas. Dakar Med. 1991;36:94-104.
- SUZUKI J, TAKAKU A. Nonsurgical treatment of chronic subdural hematoma. J Neurosurg. 1970;33:548-553.
- SVIEN HJ, GELETY JE. On the surgical management of encapsulated subdural haematoma. A comparison of the results of membranectomy and simple evacuation. J Neurosurg. 1964;21:172-177.
- TABADDOR K, SHULMAN K. Definitive treatment of chronic subdural haematoma by twist-drill craniostomy and closed system drainage. J Neurosurg. 1977;46:220-226.
- TAUSSKY P, FANDINO J, LANDHOLT H. Number of burr holes as independant predictor of postoperative recurrence in chronic subdural haematoma. Br J Neurosurg. 2008;22(2):279-282.
- WAKAI S, HASHIMOTO K, WATANABE N, INOH S, OCHIAI C, NAIGA M. Efficacy of closed-system drainage in treating chronic subdural hematoma: a prospective comparative study. Neurosurg. 1990;26(5):771-773.
- WATANABE S, SHIMADA H, ISHII S. Production of clinical form of chronic subdural hematoma in experimental animals. J Neurosurg. 1972;37:552-561.
- WEIR BKA. The osmolality of subdural haematoma fluid. J Neurosurg. 1971;34:528-533.
- YAMASHIMA T, YAMAMOTO S. The role of endothelial gap junctions in enlargement of chronic subdural hematomas. J Neurosurg. 1983;59:298-303.




